5.9. The evolution of complex life#
Professor: Nick Butterfield (Department of Earth Sciences)
Learning objectives:
Eukaryotes, animals and a planetary revolution - the Proterozoic-Phanerozoic record
Recognizing and evaluating the early evolution of eukaryotes in the fossil record
Reinventing the planet - the Cryogenian to Cambrian rise of animals
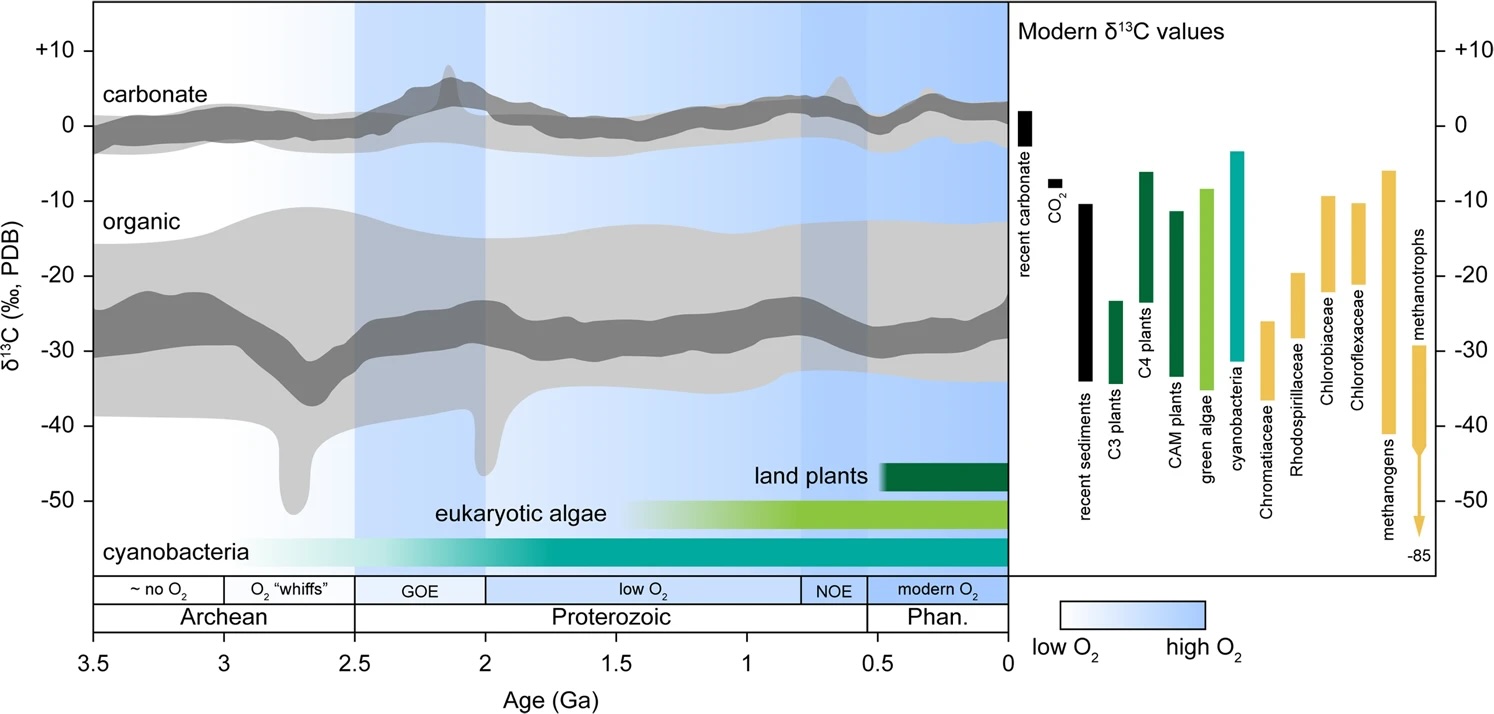
Fig. 5.43 A record of the carbon isotope composition of marine carbonates and organic matter over Earth history. The carbonate record shows remarkable constancy, despite biological innovation (photosynthesis, eukaryotes, land planets…) and major tectonic and environmental upheaval. Figure from Garcia et al. (2021).#
Mesoproterozoic: boring billion?#
As a physically habitable planet, the Earth was looking decidedly modern by at least the end of the Palaeoproterozoic (1.6 Ga). Modern-style plate tectonics were fully in place, Superior-type BIF deposition had run its course, and atmospheric oxygen was high enough to sustain both a permanent ozone layer and a modest diversity of (aerobic) eukaryotes. Curiously, these seemingly fertile new circumstances were not accompanied by much in the way of larger scale evolutionary/environmental change. The subsequent Mesoproterozoic era (1600–1000 Ma) is famous for its stable climate, monotonous redox geochemistry and excursionless \(\delta^{13}\)C values – the ‘boring billion.’
There are some signs of movement, however. The Mesoproterozoic saw the all-time high in global stromatolite ‘diversity’ and, towards its end, the first appearance of demonstrably crown-group eukaryotes. There are two key fossils to know about here: Bangiomorpha pubescens, a filamentous red alga from arctic Canada (Butterfield 2000) and Proterocladus antiqua, a filamentous green alga from North China (Tang et al. 2020). Both owe their taxonomic identification to the preservation of (more or less) complex cell-division patterns otherwise found only in modern counterparts. Because of their placement in a known sub-group of extant eukaryotes (the Archaeoplastida) these billion-year-old fossils necessarily evolved after the LECA, thus setting a hard minimum date for crown-group eukaryotes.
Neoproterozoic#
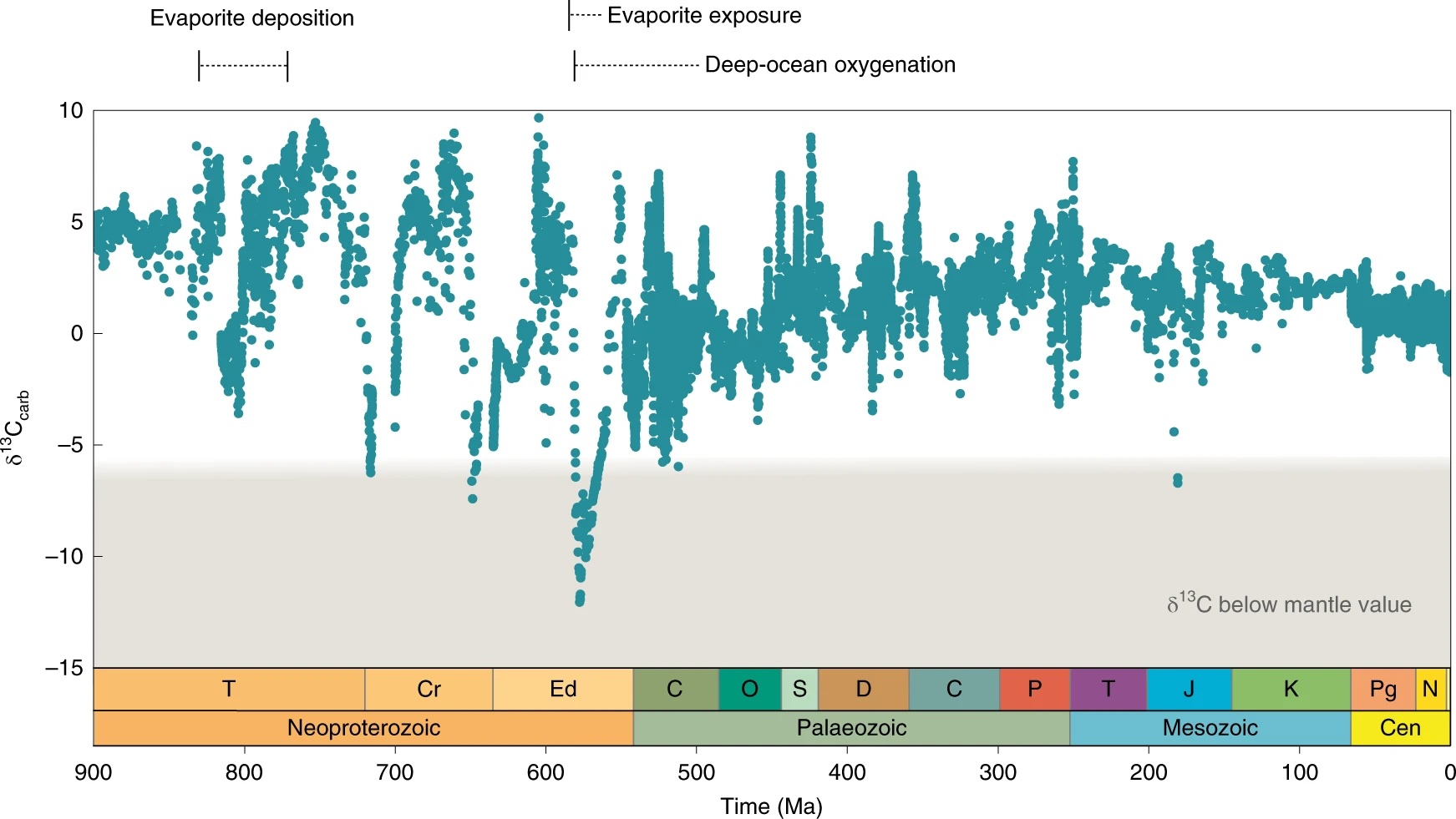
Fig. 5.44 A higher resolution record of carbonate carbon isotope compositions through the Neoproterozoic into the Phanerozoic. Figure from Shields et al. (2019).#
The pace of geobiological change picks up in the Neoproterozoic (1000–540 Ma), which is usefully divided into three parts: The Tonian (1000–720 Ma), the Cryogenian (720–635 Ma) and the Ediacaran (635–540 Ma). The Tonian sees a broad increase in \(\delta^{13}\)C values, interrupted by increasingly pronounced negative excursions at around 800 Ma – potentially associated with the breakup of the supercontinent Rodinia. Palaeontologically, the mid-late Tonian also saw the appearance of a new range of ‘armoured’ microfossils (‘testate amoebae’ and phosphate biomineralizing ‘scale microfossils’) and the first detectable record of fossil steranes (i.e., fossil biomarker molecules derived from eukaryotic sterols). At around the same time, data from some (but not all) geochemical redox proxies point to a general increase in oxidation potential, giving rise to the concept of a ‘Neoproterozoic Oxidation Event’ or NOE. And then the entire system tipped into a ice age, the first significant cold snap in more than a billion years. There is much debate over the cause and extent of these Cryogenian glaciations, as well as their broader-scale implications. In addition to the ice itself, they were accompanied by dramatic shifts in carbon cycling (δ13C), a short-lived return to BIF deposition and, at the end, emplacement of an anomalous ‘cap carbonate’ – possibly the after-effect of a ‘snowball Earth’ event.
Palaeontological data are understandably sparse through the Cryogenian, but reappear in the early Ediacaran, where both microfossil and biomarkers document a fundamental shift towards more diverse, eukaryote-dominated marine productivity. By the middle Ediacaran (~570 Ma), these are joined by the first ever appearance of macroscopic organisms in the fossil record. As ever, there is much debate over the interpretation of these Ediacaran macrofossils, but there is a compelling case for recognizing many, possibly most of them as ‘total-group’ animals.
Co-Evolution of Life and Environment#
So how do all of these geological and palaeontological data link together – or is it all just some stochastic coincidence? There is no shortage of ideas, but testability is another matter. One thing to keep in mind are the differing ecological and evolutionary premises that feed into this type of planetary-scale thinking. Whereas geologists and geochemists tend to consider things in terms of limiting resources, (palaeo)biologists are more inclined to take a ‘top-down’ approach – with evolutionary innovations introducing novel resources and opportunities of their own accord. That said, most of the recent thinking has focused on bottom-up, permissive environment-type explanations where biological evolution is held in check by some limiting resource such as oxygen or nutrients. Yes, the ‘NOE’ suggests a rise in oxygen (and animals do require oxygen), but it’s not immediately obvious how or why this same interval also saw one of the most extreme climate perturbations in Earth history, or how any of these circumstances might have ‘caused’ the evolution of e.g., tissue- and organ-grade multicellularity, complex behaviour, and the many other innovations that accompanied the Phanerozoic rise of animals.
In a lot of ways the models work better the other way around. Tziperman et al. (2011), for example, proposed that the Cryogenian glaciations may have been triggered by an evolutionary shift in marine plankton (from prokaryote-dominated to eukaryote-dominated), thereby altering the biological pump and marine alkalinity – leading in turn to a drawdown of atmospheric CO2 and corresponding collapse in planetary temperature. The trigger for the ‘rise of algae’ (Brocks et al. 2017) is still up for grabs, but might reasonably be viewed as a co-evolutionary consequence of newly evolving sponge-grade animals (Butterfield 2009, 2011, 2018; Lenton et al. 2014). As active suspension feeders, sponges also remove biological oxygen demand from the water column leading to oceanic ventilation, a phenomenon easily mistaken for atmospheric oxygenation. In other words, a late Tonian appearance of animals might be the cause (rather than the consequence) of all those bizarrely coincident perturbations marking the end of the Proterozoic.
The phenomenon of organisms influencing physical environments is known as ‘ecosystem engineering.’ Cyanobacteria clearly ‘engineered’ the Earth’s early oxygen-rich atmosphere (in concert with the tectonically driven rock cycle), and there is a comparable case for recognizing the equally disruptive impact of newly introduced animals at the end of the Proterozoic – from the construction of wave-breaking reefs and shell-beds to sediment bioturbation and fundamental re-engineering of the carbon cycle via trophic repackaging, material redistribution and the ‘top-down’ co-evolutionary reinvention of marine productivity. All that’s needed to round it off is an explanation for the (curiously belated) evolution of animals themselves. Rather than searching for some stubbornly cryptic external trigger, it is worth considering the inherent complexity of animals themselves, and the likelihood of them arising through blind evolutionary tinkering, even in a perfectly supplied lab (vanishingly small). Like cyanobacteria and plants, animals are an evolutionary singularity that dramatically reengineered planetary expression after some 3500 million years of playing by different rules.
References#
Bar-On YM, et al. 2018. The biomass distribution on Earth. PNAS 115, 6506–6511.
Butterfield NJ. 2015. The Neoproterozoic. Current Biology 25, R859–R863.
Mills DB, et al. 2014. Oxygen requirements of the earliest animals. PNAS 111, 4168–4172.
