4.3. Fundamentals of organic chemistry III: How molecules react#
Professor: Jonathan Goodman (Yusuf Hamied Department of Chemistry)
Learning objectives:
Reactions
Curly arrows
Proton transfer
Reaction energy profiles
Some molecular processes for life
Recap#
In the first lecture we looked at the properties of atoms and how they stick together to make molecules. In the second lecture, we discussed the structures of molecules, how they might be identified and how their data can be manipulated. In this final lecture, we will consider how molecules react.
Atomic orbitals interact to form molecular orbitals. Fig. 4.26 illustrates the orbitals for a hydrogen molecule and a lithium hydride molecule.
If just the bonding orbitals are filled, the energy is lowered by bringing the atoms together and so the molecule forms. If there were enough electrons to fill the anti-bonding orbitals too, the molecule would dissociate, as the \(\sigma*\) is raised in energy relative to the atomic orbitals slightly more than the \(\sigma\) bonding orbital is lowered in energy. The Highest Occupied Molecular Orbital is labelled as the HOMO and the Lowest Unoccupied Molecular Orbital is labelled as the LUMO. The difference in energy between the atomic orbitals and the molecular orbitals is greater for hydrogen than for lithium hydride, both because the atomic orbitals are closer to each other in energy and also because they are closer to each other in size. For lithium hydride, the HOMO is closer in energy to the hydrogen atom than to the lithium atom, and so the electrons are closer to the hydrogen. This polarises the lithium-hydrogen bond so that the lithium atom has a partial positive charge and the hydrogen a partial negative one.
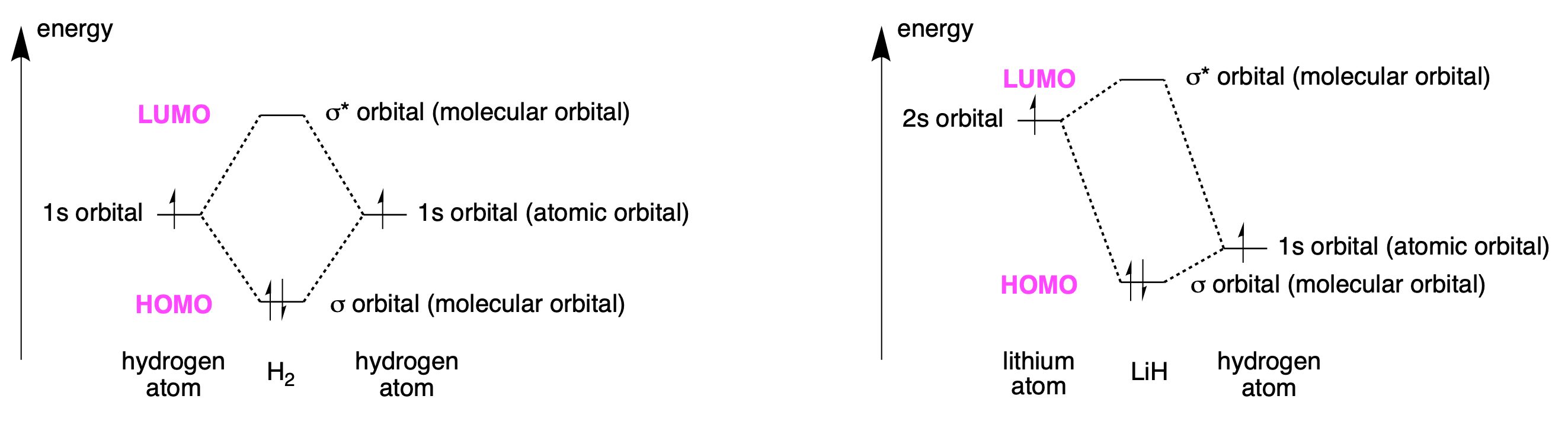
Fig. 4.26 Hydrogen (left) and lithium hydride (right). For both, the two atomic orbitals combine to form two molecular orbitals, one bonding and one anti-bonding.#
The figure gives an account of why these atoms stick together and of some of the properties of the molecules. There are two ways to break the bond between the atoms: either remove the electrons from the bonding orbital, or add electrons to the antibonding orbital. When two molecules interact, the electrons from the HOMO of one may move into the LUMO of the other, thus causing the bonding of the molecules to change. This is a reaction happening.
Describing reactions wih curly arrows#
For many molecules, the electrons are present in pairs, and a reaction corresponds to the movement of a pair of electrons. This can be indicated by drawing a curly arrow with a full head. Fig. 4.27 gives examples of this. The movement of a single electron is indicated by a curly arrow with half a head.
In the left hand process, a curly arrow starts at a negative charge. A single electron has a single negative charge, but the curly arrow corresponds to the movement of a pair of electrons. This is consistent, because the nucleus balances one of the negative charges of the two electrons, but not both. This arrow shows how the electrons move to the adjacent bond, making a single bond into a double bond. This would give the carbon atom at the other end of the bond five bonds (two double and one single), which would be inaccessibly high in energy. The second curly arrow breaks the original double bond, and moves the electrons onto the top oxygen. These curly arrows turn the first structure into the second structure. The curly arrows on the second structure move the electrons back to re-form the first structure.
These curly arrows show the movement of electrons and they change one representation of the molecule into another. In this example, both of the representations are the same molecule. The negative charge is spread over the two oxygen atoms. Because this is not a change in the molecule, these curly arrows do not correspond to a reaction. The double-headed arrow between the structures represents resonance and not a reaction.
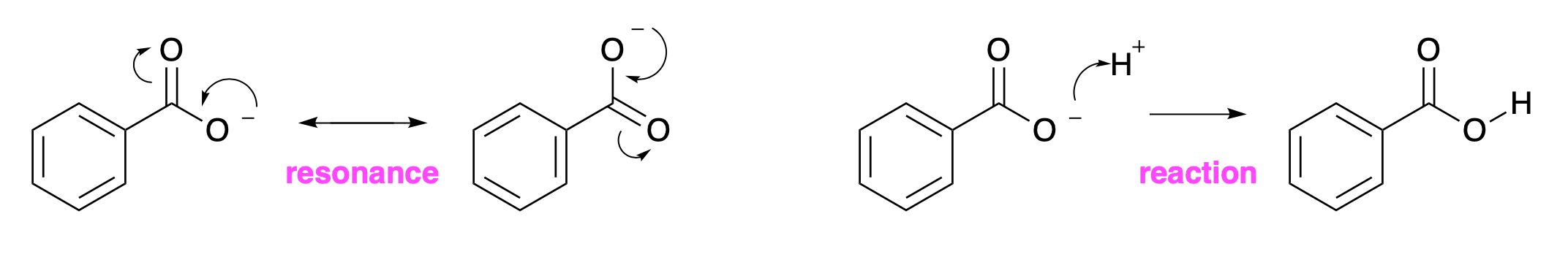
Fig. 4.27 Resonance structures for a carboxylate (left) and a reaction that protonates a carboxylate (right).#
The right hand process in Fig. 4.27 does correspond to a reaction. The negative charge on the molecule moves to a proton. This corresponds to the HOMO of the carboxylate to the LUMO of the proton, forming a new \(\sigma\) bond between the oxygen and the hydrogen.
Proton transfer#
This simple reaction is driven by a negative charge moving towards a positive charge. If there are no other molecules around, perhaps because they are outside a planetary atmosphere, this will be a strong driving force. If the molecules are dissolved in a liquid, the charge interaction may well be attenuated by the medium. A terrestrially-centred view would assume that the solvent is water, which has polar bonds and so will interact favourably with the charges. This will reduce the driving force for the reaction. If the solvent were methane, the driving force for the reaction would be closer to that without a solvent.
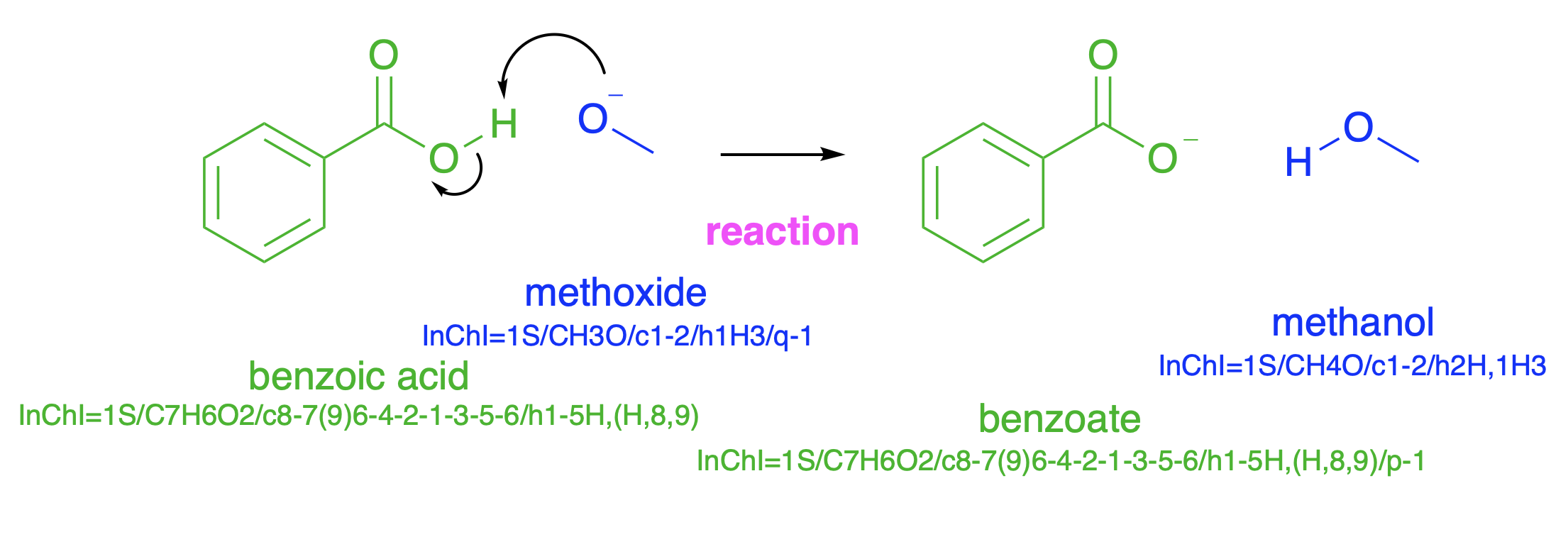
Fig. 4.28 Proton transfer from a carboxylic acid to an alkoxide. The colours are present only to link the structures to the names and to the InChI. In this example, with simple molecules, the InChI are rather longer than the names. For more complex molecules, the names can no-longer be decoded using Google or PubChem, and the InChI become easier to handle, at least for computers.#
Fig. 4.28 shows a slightly more complicated process. The negative charge on methoxide attacks the hydrogen on benzoic acid, and the product is a benzoate ion and methanol. The driving force for this reaction is conversion of a negative charge which is localised on a single oxygen atom to a lower energy negative charge spread out over two oxygen atoms, and, to a small extent, the central carbon atom (Fig. 4.27, left).
The orbitals involved in this process are shown in Fig. 4.29. The HOMO for the process is a lone pair on the methoxide anion. The LUMO is the \(\sigma*\) anti-bonding orbital of the O-H bond. The \(sp^2\) orbital on the oxygen is lower in energy than the \(1s\) orbital on the hydrogen, so the electrons in the bonding orbital (\(\sigma\)) are more around the oxygen than the hydrogen. The \(\sigma*\) anti-bonding orbital, therefore, has its largest lobe on the hydrogen, and this is where the alkoxide prefers to interact with it.
Filling the \(\sigma*\) anti-bonding orbital breaks the O-H bond. At the same time, a new bond is formed between the alkoxide oxygen and the hydrogen, so forming the product. The reaction can only happen if the reacting orbitals are lined up in a suitable way. If this reaction were to be done in solution, the molecules would move around randomly, and collide in the right orientation from time to time. Terrestrial life may often speed up these reactions by building structures out of amino acids, called enzymes, which can help molecules reach the best orientations, and also stabilise any high energy structures which may be on the reaction pathway. Life without this type of machinery is hard to imagine, although it is possible to build enzyme-like structures which are not made of amino acids.
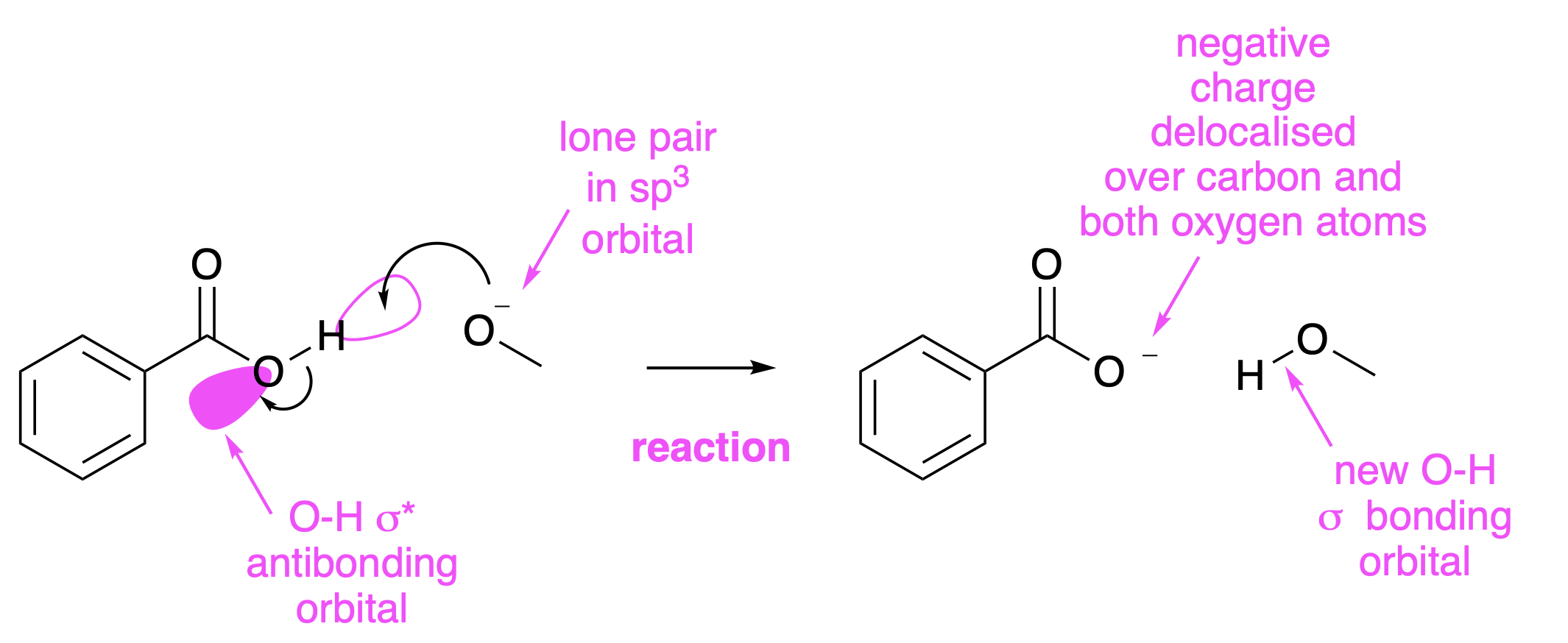
Fig. 4.29 The orbitals involved in the proton transfer process.#
Why does the reaction go in this direction? Some hydrogens are attached more firmly than others, and changing to a form with a stronger O-H bond lowers the overall energy of the system. Many such reactions have been studied, and the results are quantified as the \(pK_a\) for molecules, which is a number describing how easily a molecule can dissociate into an anion and a proton. These measurements are solvent dependent, as different solvents interact with cations and anions in different ways. Nearly all measurements of \(pK_a\) values are made in water at about \(300 K\). These are very useful for considering chemical reactions occurring in life on earth, but may be misleading for reactions happening under very different conditions.
Fig. 4.30 illustrates how energy changes as the reaction proceeds. Most reactions have an energetic barrier which needs to be overcome, the activation energy. Enzymes are able to low the barrier and so speed up reactions. The difference in energy between the starting materials and the products determines whether the reaction ends up with a mixture of starting material and product, if the energy change is small, or with nearly all of the product, if the energy change is large and favourable.
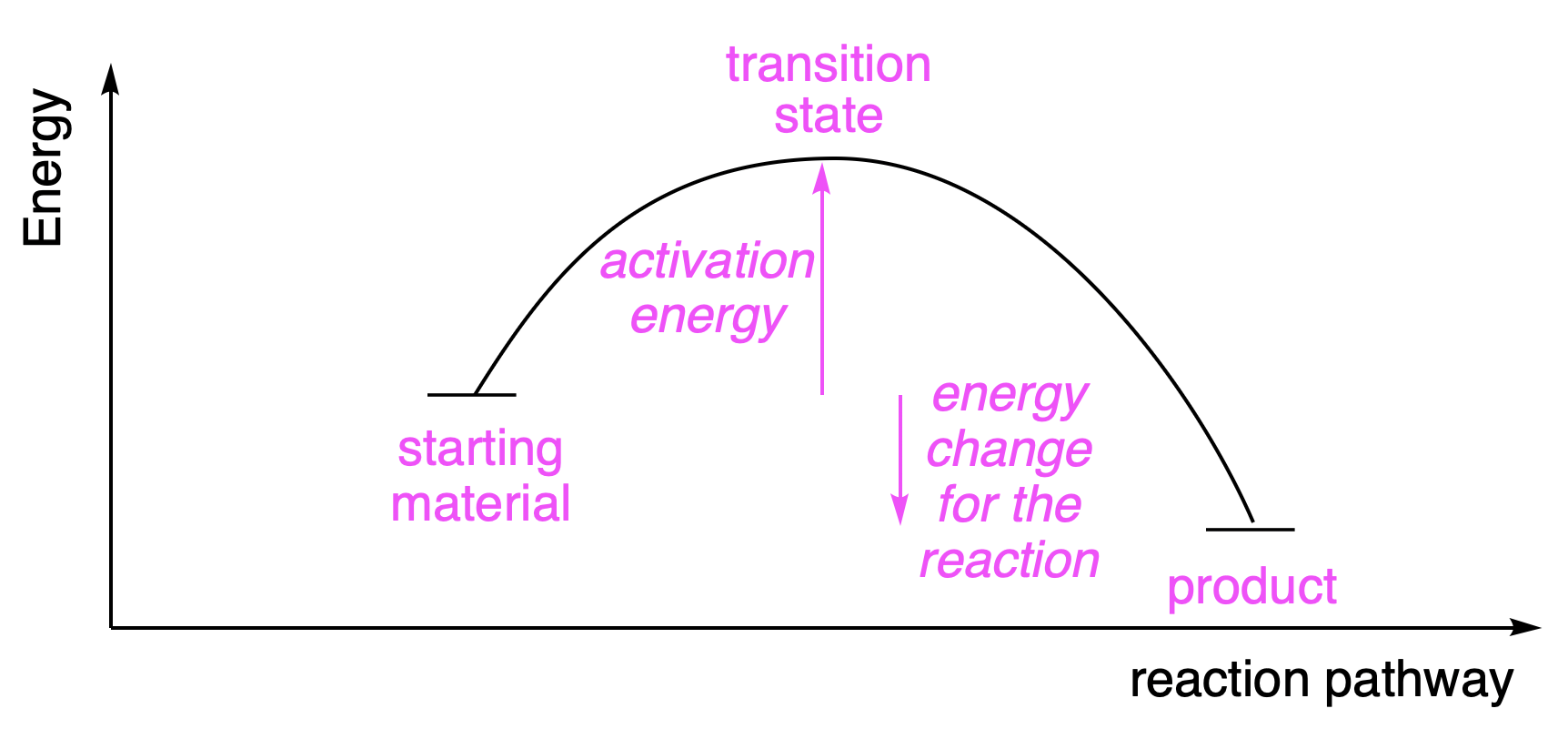
Fig. 4.30 The energy changes as reactions proceed. The rate of the reaction (kinetics) is controlled by the activation energy: reactions with low activation energies go faster. The position of the equilibrium (thermodynamics), the eventual ratio between starting materials and products, is controlled by the energy change for the reaction.#
When competing reactions are possible, which is a very common situation, the reaction which occurs to the greatest extent may be the fastest one (kinetic control) or may be the one which is most energetically favourable (thermodynamic control). Alex Thom will talk in more detail about calculations on these processes.
Amide formation from esters#
Proton transfer is an important process, but does not make new bonds between second row elements. Building up larger molecules by forming such bonds is probably essential to build substances with the necessary complexity to form life. Fig. 4.31 shows how an amine can react with an ester in order to make an amide.
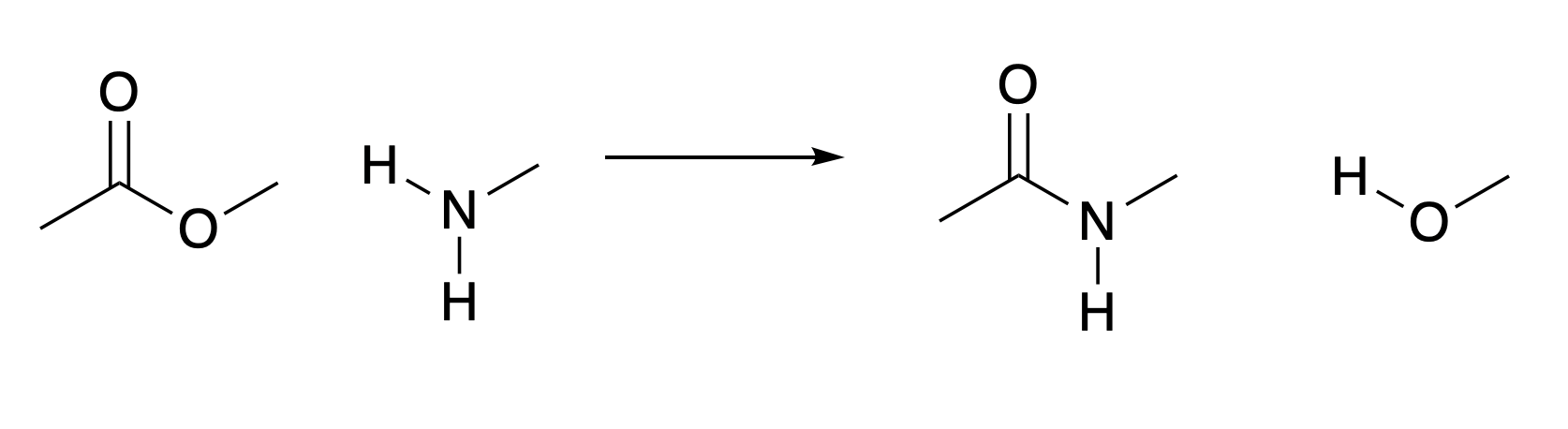
Fig. 4.31 Amide formation from an ester. This figure does not show the mechanism, but you should be able to work out which bonds break and which bonds form. The HOMO in the starting materials is the nitrogen lone pair. The LUMO is the anti-bonding orbital of the carbon-oxygen double bond.#
The mechanism for this reaction probably has a number of steps. The HOMO will interact with the LUMO but this does not lead directly to the product. A fuller version of the mechanism is given in Fig. 4.32. All six steps must take place, although mechanisms are not customarily drawn in such detail. Protons adding and dropping off are straightforward steps, provided that the environment makes protons available. If the reaction were to be enzyme-catalysed, then a large enzyme molecule surrounding the reaction might put protons, or other systems with similar properties, in just the right place for the reaction to occur smoothly
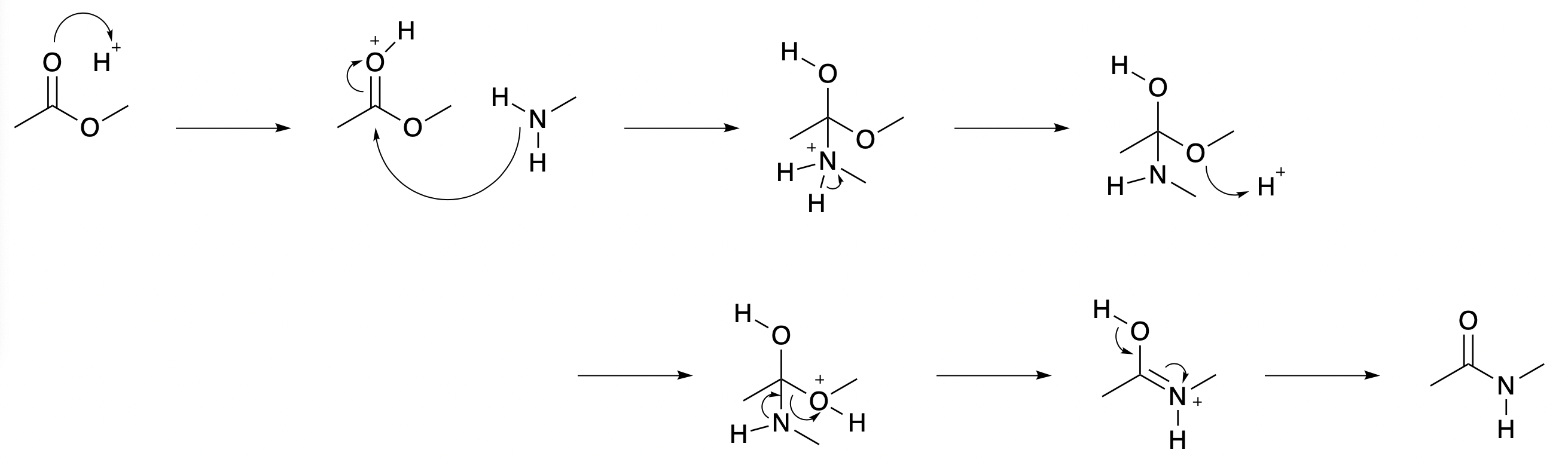
Fig. 4.32 Amide formation from an ester. All of these steps need to happen and all have their own HOMO / LUMO interactions. I have assumed that protons (\(H^+\)) are available. If the solvent is water, this should be the case.#
Joining up amino acids#
Enzymes and other key molecules for terrestrial life are built up from chains of amino acids. At the beginning of lecture two, we looked at alanine, which is a simple amino acid which is naturally-occurring on earth. There are about twenty main naturally-occurring amino acids, differing in their sidechain. Alanine has a methyl group as its sidechain and other groups can be placed here.
Fig. 4.33 shows four alanine molecules as the starting materials, which can join up by forming amides, in a similar way to Fig. 4.31, to form a chain of four amino acids. The four alanine molecules which are the starting materials have all been drawn in different, reasonable ways, and all represent the same molecule. It is not immediately obvious that this is the case. It can be checked by generating the InChI of all of the structures, which is identical for all four.
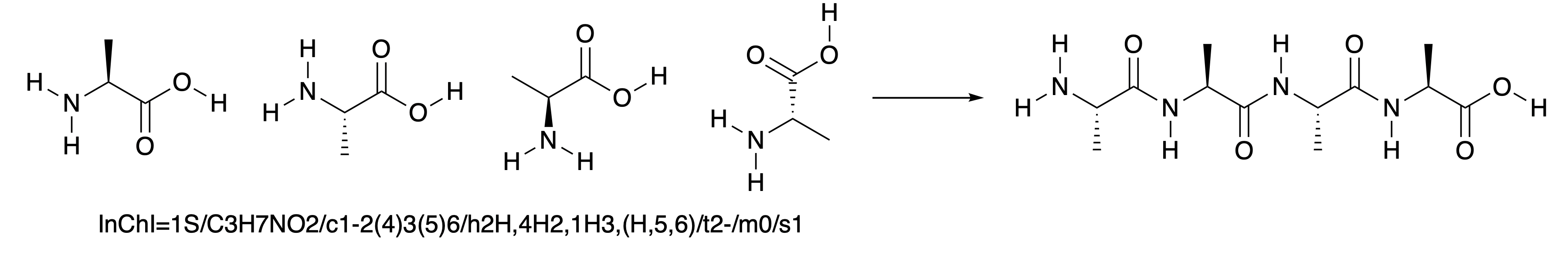
Fig. 4.33 Four alanine molecules, joining up to form a chain.#
The mechanism of the reaction could be drawn in a similar way to Fig. 4.32 only three times as long. However, there is a problem. If alanine is dissolved in water, or other solvent which allows protons to move around the molecule as required by the mechanism, a proton will move from the right hand side to the left hand side (Fig. 4.34). This means that the nitrogen atom no longer has a lone pair to act as the HOMO in the reaction, and the \(\pi*\) LUMO on the carbon-oxygen double bond is close to a negative charge and so much less accessible to incoming electrons. How is it possible for amino acids to join together?
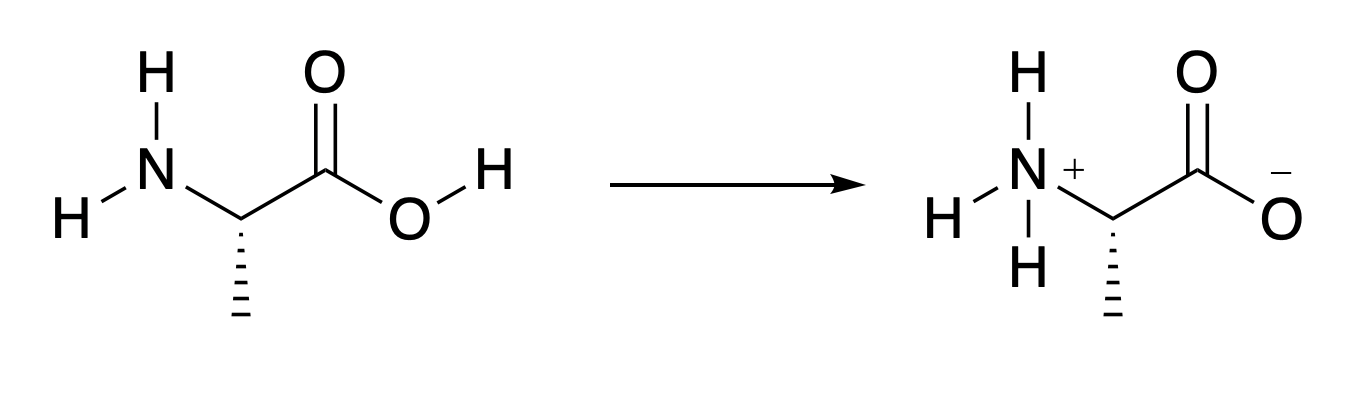
Fig. 4.34 An alanine molecule in water.#
In terrestrial amide formation, enzymes are available which allow this transformation to happen. The details of the pathway are even more complex that Fig. 4.32. Fortunately for terrestrial life, this process works very well. However, it leads to an important question: if we need the molecules of life to make the molecules of life, how did life begin? These three lectures are insufficient to address this rather fundamental problem. Extra-terrestrial life must have had a way of addressing this issue too.
Making carbon-carbon bonds#
Carbon-carbon bonds are central to terrestrial life. How do they form? The carbon-carbon bonds in you (there are a lot) are mainly formed by aldol reactions, Fig. 4.35. The example illustrated is the simplest possible aldol reaction, and there are many more complicated variants and some of these are used in life on earth. The example shows the idea. The proton adjacent to the carbon-oxygen double bond can fall off, because the electrons from the C-H \(\sigma\) bond can move into the vacant \(\pi*\) antibonding orbital, transforming the carbon-oxygen double bond into a carbon-oxygen single bond. This generates a molecule called an enol which can then attack another molecule of the starting material. This creates a new carbon-carbon bond, building up molecules containing more carbon atoms. This is the process that creates most natural carbon carbon bonds on earth.
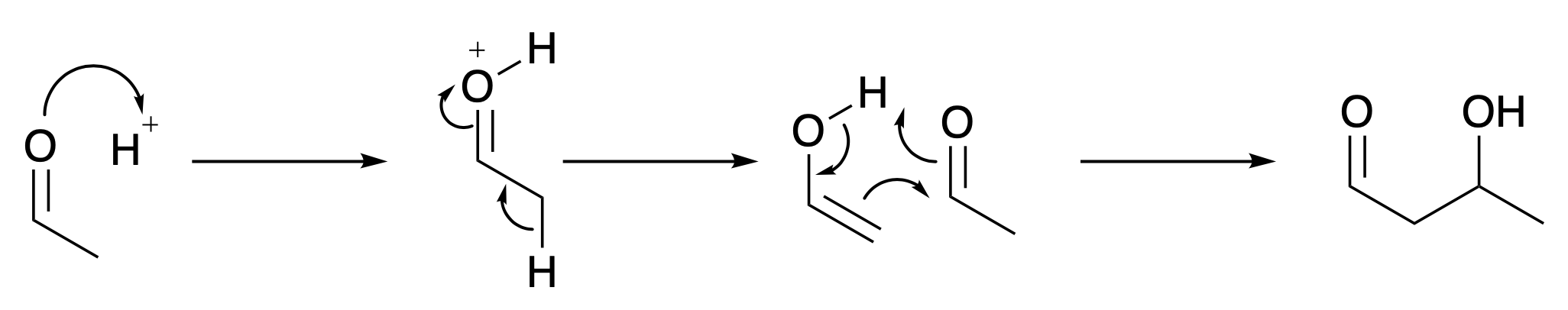
Fig. 4.35 The aldol reaction.#
Summary#
Atoms we can understand quite well, at least for first two rows of the periodic table: they are squashy, sticky spheres which interact in complex ways. Their behaviour can be understood, reasonably well, by thinking about 1s, 2s and 2p orbitals, and how they can hybridise so they interact in pairs to form larger molecules.
These molecules can be ordered and catalogued, and their properties calculated rather precisely
They can react in complex ways to form many, many more molecules.
What I have told you covers the basics of the majority of life on earth. Other elements are needed for life, particularly phosphorous and some non-lithium metals, but what I have told you is enough to make most proteins, DNA/RNA (except for the phosphorous in the backbone), and the majority of natural products.
Life on other planets is likely to challenge the assumptions we make, sometimes subconsciously. \(300 K\) is a good temperature, \(100 kPa\) is a good pressure, water is a common liquid, combusting sugars with oxygen is a useful energy source, amino acids are convenient building blocks for biomolecules, DNA is a good way of storing information. All of these things are true on earth, but may not be true on other planets. Life in the universe could be very diverse, but the properties of atoms and how they build into molecules will be the same everywhere.
