5.7. Early life on Earth#
Professor: Nick Butterfield (Department of Earth Sciences)
Learning objectives:
Overview of the history of life on Earth
Interrogation of early ‘biosignatures’
Evidence of early life on Earth
Coevolution of life and the planet
Early life on Earth: interrogating the data#
As far as we know (so far), ours is the only body in the Universe to support a biosphere. What’s more, it appears to have done so for quite a long time. There is a rich fossil record extending back through the Phanerozoic Eon – the last 540 million years of Earth history – and a more cryptic record through the preceding Proterozoic (2500 to 540 million years ago) and Archean (4000 to 2500 Ma) eons. There are no surviving Hadean (> 4 Ga) rocks on Earth, which introduces some obvious, but not necessarily insurmountable, challenges to reconstructing a full record.
Age (Ga) |
Archive |
|
|---|---|---|
Solar system |
4.567 |
CAIs |
Earth |
4.5 |
Terrestrial Pb deposits |
Moon |
4.5 |
Lunar samples |
Oldest Earth minerals |
4.4 |
Jack Hills zircons, Western Australia |
Oldest rocks |
4.03 |
Acasta Gneiss, Slave Province Canda |
Oldest metasediments |
\(>\)3.7 |
Isua Supracrustal Belt, SW Greenland |
Oldest well preserved seds |
3.5 |
Pilbara & Swaziland Supergroups |
End of intense bombardment |
~3.9 |
Lunar cratering |
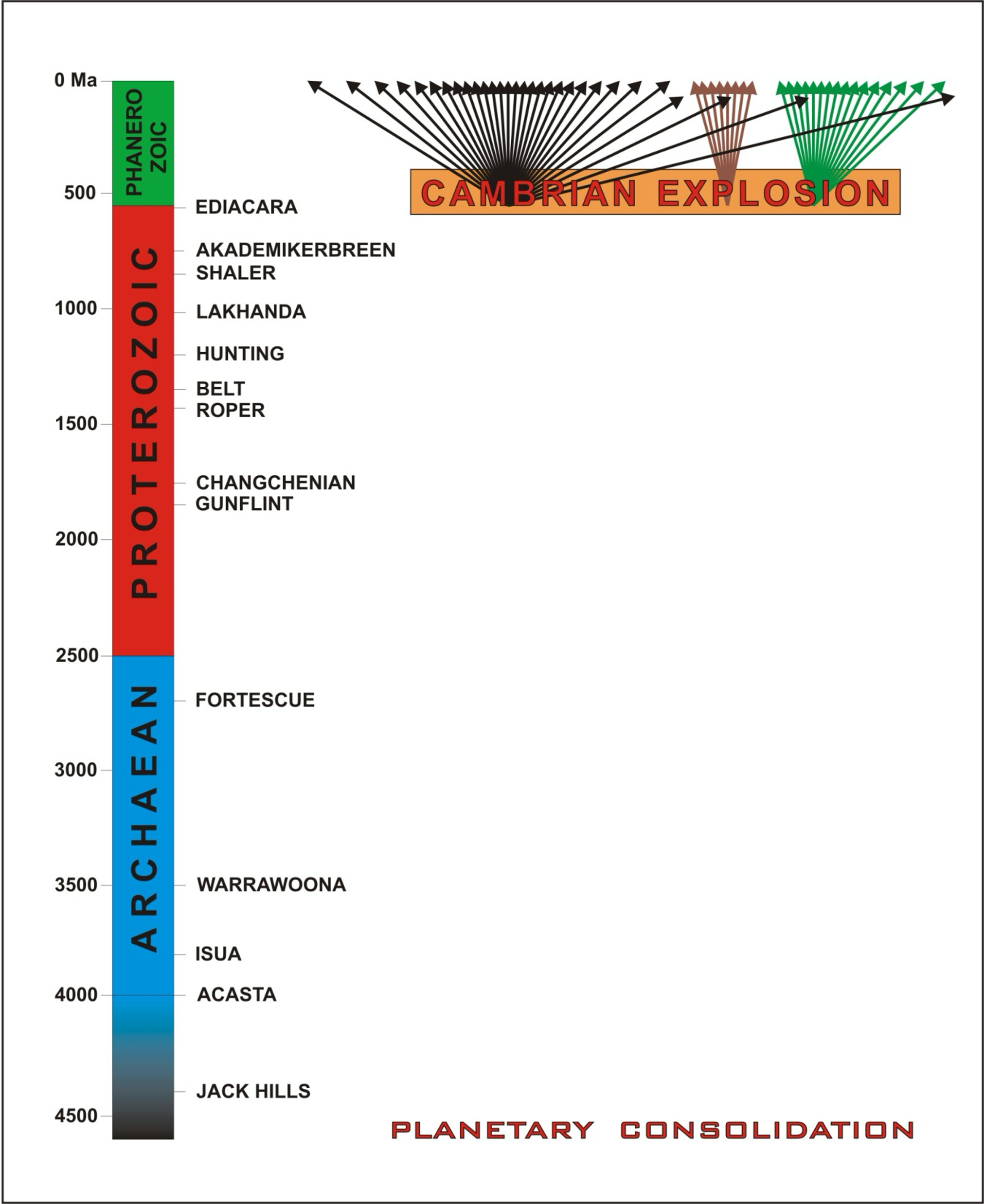
Fig. 5.35 The Geological Time Scale and rock units of significance in piecing together the deep-time record of life on Earth. Reconstructing the sequence of events between ‘planetary consolidation’ and the Cambrian Explosion is a fundamental pursuit in its own right, but also a prerequisite for any meaningful search for life on other worlds.#
There are various ways of addressing the evolutionary history of life on Earth, but the only direct approach is via the fossil record. For our purposes, fossils can be defined as any evidence of past life preserved in the rock record, primarily (but not exclusively) in sedimentary rocks formed at the Earth’s surface. Fossils come in three basic flavours:
Body fossils – anatomically preserved remains or equivalent representations of once-living organisms. In the Precambrian (i.e. pre-Phanerozoic) record, these will be represented almost exclusively by microfossils. There are all sorts of pitfalls however, not least in distinguishing true microfossils from more recently introduced microbes/contamination, and from diagenetic artifacts that can mimic microbial forms. In other words, demonstrating syngenicity and biogenicity.
Trace fossils – sedimentary structures resulting from the presence and activity of once-living organisms. Classic examples include the burrows and borings of (Phanerozoic) animals, but the earlier record is dominated by the effects of microbial mats on sedimentation, primarily via the trapping and binding of sedimentary grains on exposed surfaces and/or the biologically induced precipitation of early diagenetic cements associated with microbial metabolism.. Fossilized occurrences of this collective activity are known as microbialites and are best known from regularly-layered accretionary stromatolites (vs. clotted-fabric thrombolites). As with body fossils, there are issues with distinguishing bona fide biogenic microbialites from superficially similar abiotic structures such as speleothems and diagenetic/tectonic artifacts. Some skeletal organisms with accretionary growth also end up looking disturbingly stromatolitic.
Chemical fossils – (geo)chemical signatures derived from once-living organisms, or as a by-product of their metabolic activities. The standard examples are taphonomically recalcitrant lipid biomarker molecules extractable from organic-rich sediments; e.g., dinosterane from dinoflagellates. More generally, evidence of past life can be found in distinctively biogenic isotopic fractionations, or redox signatures associated with conditions substantially beyond thermodynamic equilibrium. Evidence of an oxygen-rich atmosphere, for example, could be considered a ‘chemical fossil.’ As ever, there are first-order concerns with secondary alteration, contamination and interpretation.
Microbial relationships and phylogeny#
All extant organisms share the same basic chemistry and cellular machinery – clear evidence that all extant life has descended from a single last common ancestor. Deep phylogenetic relationships can potentially be resolved by comparing homologous features, particularly the macromolecular sequences that constitute nucleic acids and proteins. When it comes to resolving the overall structure of the universal tree of life, the molecule of choice is small subunit ribosomal RNA (SSU rRNA) because it:
is universally distributed
has a constant function (protein synthesis)
is highly conserved/slowly evolving
is large enough to provide phylogenetic information
is not subject to substantial horizontal gene transfer
SSU rRNA (along with various other molecular, cellular and physiological features) points to the presence of three fundamental Domains of life:
Archaea (= ‘archaebacteria’) are represented by a metabolically and ecologically diverse range of prokaryotic (i.e., non-eukaryotic) microbes, including methanogens, haloarchaea and various hyperthermophilic (> 80°C) and acidophilic forms. In addition to their distinct genetic signatures, the Archaea are united by the distinctive ether lipids of their cell membranes.
Bacteria (= ‘eubacteria’) are represented by around 25 ‘phyla’ of prokaryotic microbes. This phylogenetic diversity is matched by a comparable range of metabolic and ecological habits, but are generally less ‘extremophilic’ than Archaea, and do not include methanogens. On the other hand, they are the only domain of life to have evolved true photosynthesis, both oxygenic (cyanobacteria) and anoxygenic (including chloroflexus, purple bacteria, green sulfur bacteria, and heliobacteria).
Eukarya (= eukaryotes) include the three multicellular/tissue-grade Kingdoms (plants, animals and fungi), along with a massively diverse range of unicellular and multicellular (but sub-tissue-grade) protists. The domain includes no known hyperthermophiles, or even thermophiles. As eukaryotes evolved from within Archaea, strictly speaking there are only two truly fundamental domains of life…
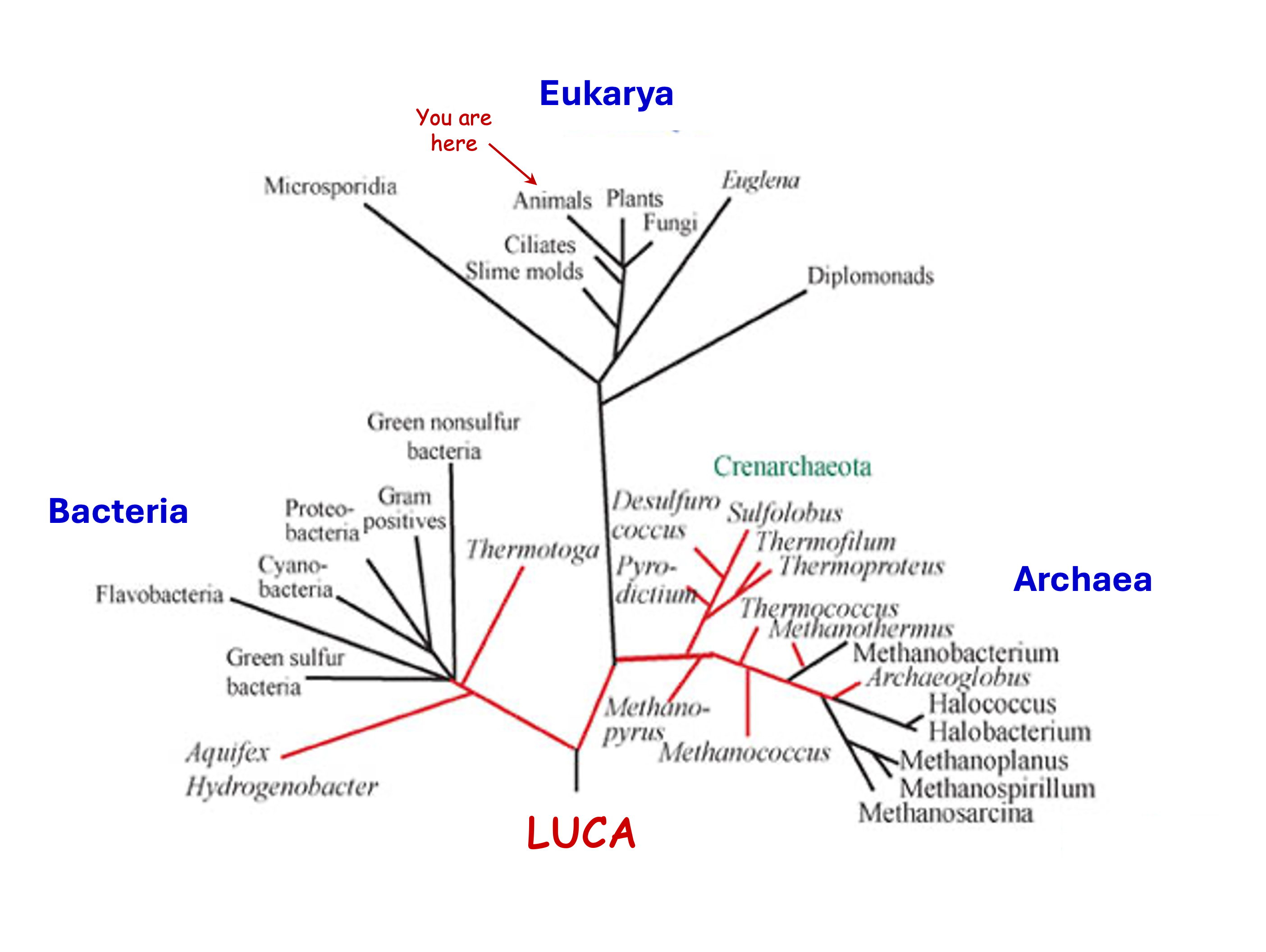
Fig. 5.36 A ‘Universal Tree of Life’ based on SSU rRNA showing the three fundamental Domains of life and their phylogenetic roots leading back to LUCA - the Last Universal Common Ancestor.#
Microbial ecology/metabolism#
Carbon-based life requires a replenishable source of both energy (ultimately in the form of electrons) and carbon. There is a useful set of terms for describing the underlying metabolic strategies practiced by different organisms as they capture and recycle these resources:
If… |
…then the organism is… |
|---|---|
the C source is CO\(_2\) |
autotrophic |
the C source is organic C then |
heterotrophic |
the energy source is light |
photo-trophic |
the energy source is chemical |
chemo-trophic |
the energy source is inorganic |
litho-trophic (e.g., chemolithoautotrophic) |
the energy source is organic C |
organo-trophic (= heterotrophic) |
Many microbes are capable of both photosynthesis and heterotrophy; i.e., mixotrophic. Some are photoheterotrophic, deriving some of their energy from solar radiation (capturing and storing it as ATP), but unable to fix/synthesize their own reduced carbon.
Stable Isotope Fractionation#
Carbon has two important stable isotopes, \(^{12}\)C and \(^{13}\)C. The \(\delta^{13}\)C notation indicates the \(^{13}\)C/\(^{12}\)C ratio of a sample relative to that of a standard in parts per thousand. \(\delta^{13}\)C represents the isotopic fractionation between the biologically fixed organic carbon (Corg) and ‘abiotically’ precipitated carbonate carbon (Ccarb)
Due to kinetic isotope effects during carbon fixation, \(^{12}\)C is preferentially incorporated into organic carbon during photosynthesis or chemosynthesis. There are a number of biochemical pathways for carbon fixation, each of which imparts a characteristic range of carbon isotope values. By far the most common, at least in the modern biosphere, is the Calvin-Benson cycle, which is based on the enzyme Rubisco (ribulose-bisphosphate carboxylase/oxidase). Rubisco is directly responsible for a ~25‰ fractionation in \(\delta^{13}\)C, with another 8‰ contributed by temperature effects – giving a maximum \(\delta^{13}\)C of 33‰. Significantly, there is a stratigraphic record of isotopically depleted organic carbon – and correspondingly enriched carbonate carbon – extending back to the early Archean. This 25–30‰ difference is arguably the most convincing evidence for an active early Archean biosphere.
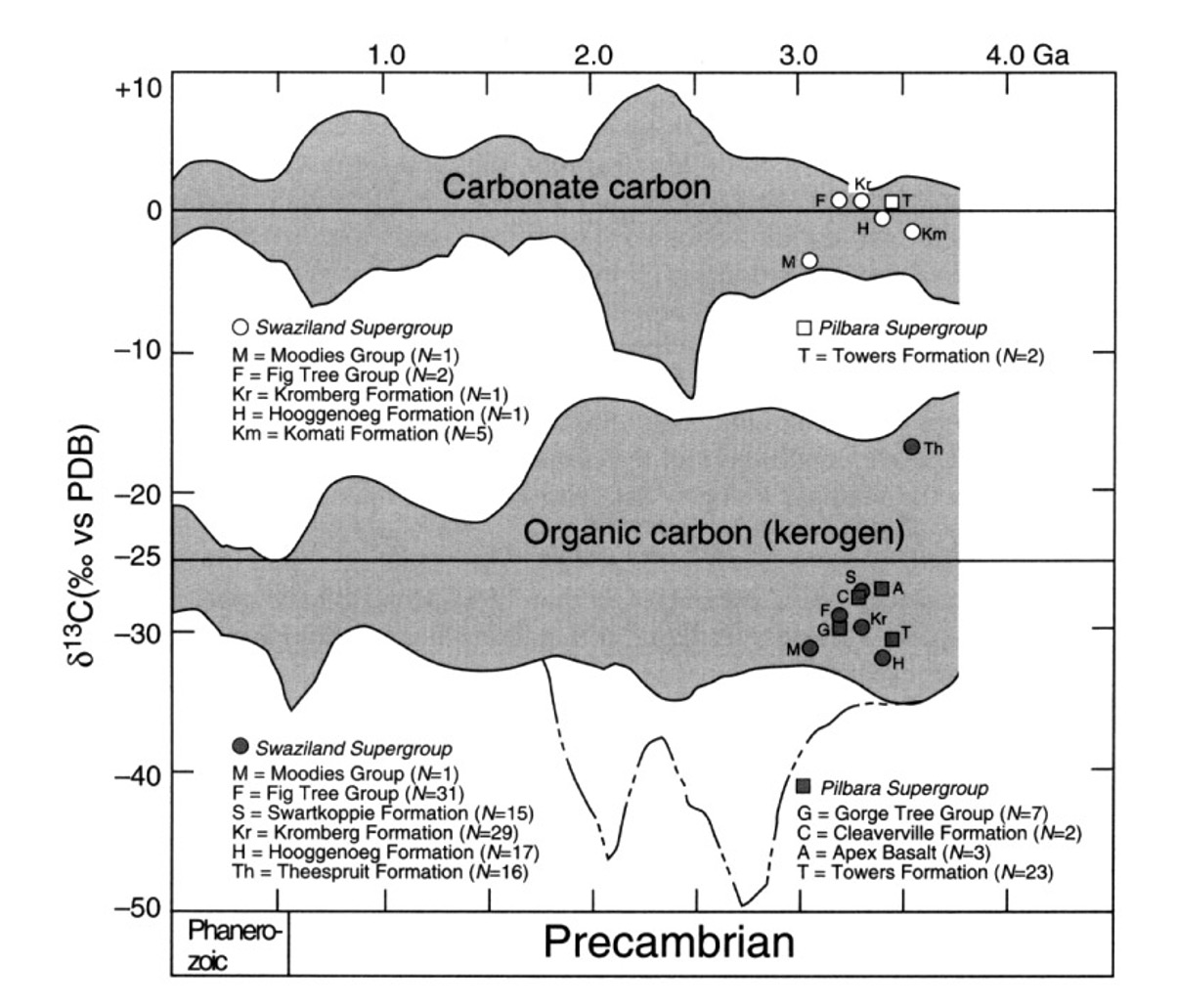
Fig. 5.37 The carbon isotope composition (\(\delta^{13}\)C) of marine sediments over the past 3.5 billion years, illustrating the pronounced fractionation between carbonate and organic carbon - compelling evidence for the presence of an active carbon-based biosphere.#
Sulfur has two principal stable isotopes, \(^{32}\)S and \(^{34}\)S. The \(\delta^{34}\)S notation indicates the ratio of a sample relative to that of a standard in parts per thousand. In marine sediments, \(\Delta\delta^{34}\)S represents the isotopic fractionation between seawater sulfate and sulfide and is mediated by various types of sulfur-metabolizing microbes.
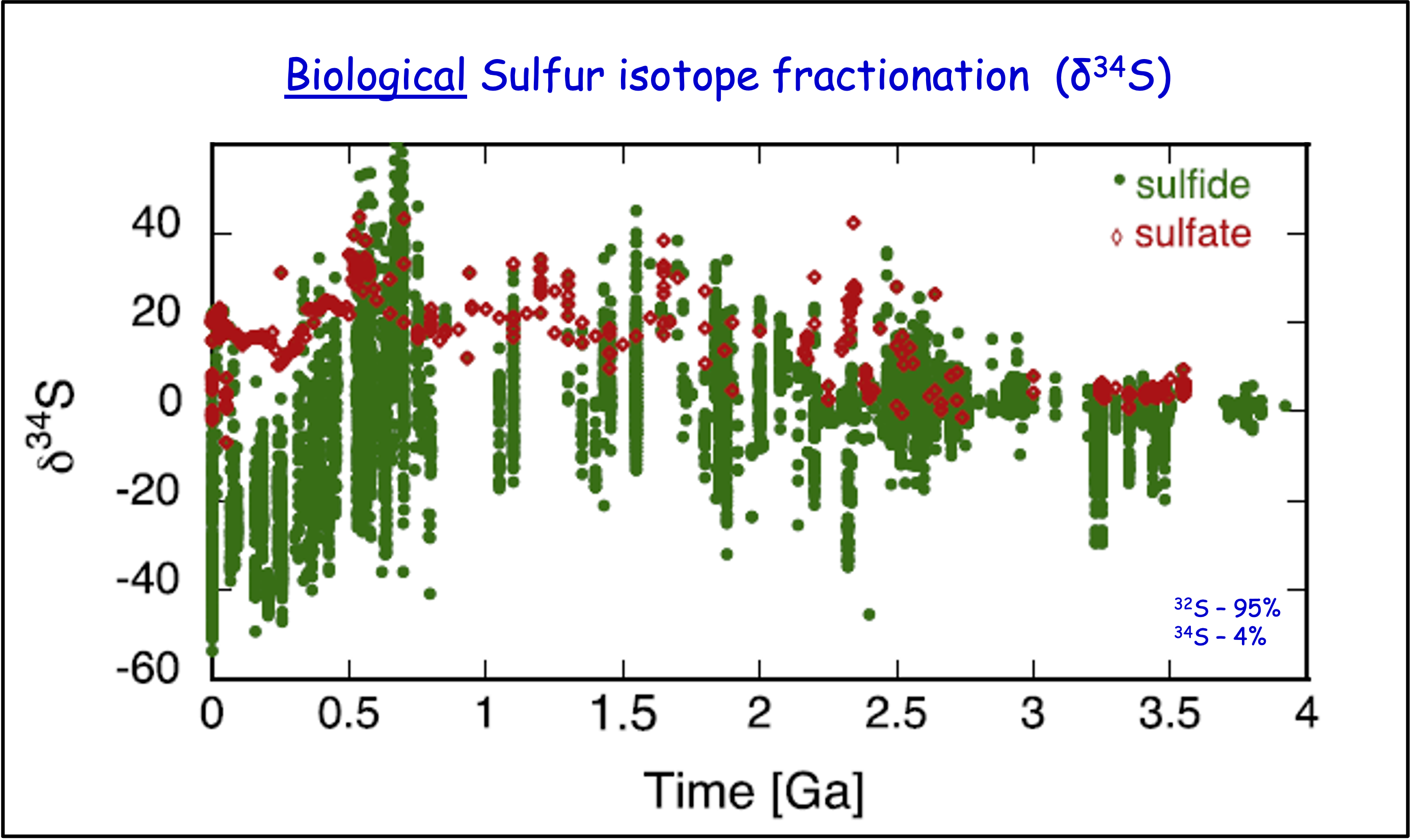
Fig. 5.38 The sufur isotope compositin (\(\delta^{34}\)S) of marine sediments over the past 3.5 billion years, documenting a long-term increase in fractionation between sulfate and sulfide sulfure - evidence of increasingly active (biologically mediated) sulfur cycle.#
Sulfate reducing bacteria (SRB) preferentially metabolize \(^{32}\)S resulting in isotopic fractionations of 40–45‰, but only where sulfate is non-limiting. Low levels of fractionation imply low sulfate concentrations – or potentially an absence of SRB. The presence of low but measurable \(\delta^{34}\)S fractionation in early Archean sulfates/sulfides points to the early establishment of SRB.
Early Archean environments#
Fossils are preserved in rocks, and it is important to appreciate that this geological archive disappears by around 4 Ga – a consequence of (external) weathering and (internal) tectonic cycling. At the same time, the signal that does survive is further confused by the non-uniformitarian nature of the processes themselves. The early Archean atmosphere, for example, contained no significant oxygen, under a sun that was 30% less luminous than today’s, and on a planet that had yet to differentiate most of its continental lithosphere. The current textbook view holds that modern-style plate tectonics only fired up in the later Archean, with the early Archean represented largely by Granite Greenstone Belts. The supracrustal greenstones are represented by huge thicknesses of basalts, within which are slivers of sedimentary successions – and the primary focus of attention in the search for the earliest life on Earth. The best preserved of these are on the Pilbara craton, NW Australia and the Kaapvaal craton (Barberton Greenstone Belt) in southern Africa. The oldest is the ~ 3.8 Ga Isua Greenstone Belt in SW Greenland.
Based on a combination of (possible) microfossils, (probable) microbialites, and carbon isotope data (\(\Delta\delta^{13}\)C), there is a compelling case for recognizing the presence of life on Earth by at least 3.5 Ga – and a reasonable one for extending this to ~ 3.8 Ga. The rest is just history – a co-evolutionary interplay of organisms and environment.
References#
Boehnke P, Harrison TM. 2016. Illusory late heavy bombardments. PNAS 113:10802–10806.
Javaux, E.J. 2019. Challenges in evidencing the earliest traces of life. Nature 572, 451–460.
Valley JW, et al. 2002. A cool early Earth. Geology 30:351–354.
