3.11. Venus I: Venus Today#
Professor: Paul B. Rimmer (Physics)
Co-Author: Arwen E. Rimmer
Learning Objectives:
This course is about Venus as it is today. It will cover:
History of Venus observation: lander, probe, satellite, ground-based.
Data about the atmosphere, clouds, surface and subsurface
Mysteries about Venus’s atmosphere today
The best models applied to address these mysteries
Brief History of Human Thought about Venus#
Ancient History#
This is the journey of Venus: after an absence of eight days, Venus appears right on the edge of the horizon before sunrise, and over the next 263 days it travels upwards, reaching an angle of no more than 47 degrees, before falling back down. It then disappears for 50 days, appearing again on the horizon, but this time at sunset, where it proceeds to rise and fall again over another 263 days before disappearing once more. Astronomers call this 584 day cycle Venus’s synodic period, meaning it takes this long for the exact positional relationship between Earth, Venus, and the Sun to recur.
We know now this peculiar circuit is simply a result of Venus’s interior orbit, how when it passes in between us and the Sun, or behind it, the Sun’s light blots it out. And it comes out in the morning when it overtakes our orbit, and in the evening when it’s catching up. But to stargazers of long ago, these motions were mysterious, portentous, even. They saw the heavens as a macrocosm of humanity’s persistent struggles; good vs evil, men vs women, young vs old, strong vs weak, rich vs poor, modernity vs tradition. It was perhaps only natural that they interpreted Venus’s recurrent rise and fall in grandiose, mythological terminology.
Not all ancient societies realized that Venus in the morning and Venus in the evening were the same “star”. But the oldest surviving mythology built around Venus comes from the ancient Sumerians of Mesopotamia, who were sufficiently knowledgeable about astronomy to figure it out more than five thousand years ago, at the dawn of the Bronze Age (Cooley 2008).
In Ancient Sumeria, Venus’s eternal return was depicted in tales of the goddess Inana (later called Ishtar by the Babylonians), who had a dual role as the deity of both love and war. Astrological traditions link the story of Inana’s Descent into the Underworld to Venus’s celestial motions (Hostetter 1991). Therein the goddess travels to the realm of the dead, where she is killed, and then resurrected three days later to return to the heavens. This could be seen as an allegory for the planet’s recurrent periods of ‘invisibility’, when it switches from morning to evening star.
The oldest surviving astronomical text in the world comes from a later Babylonian cuneiform tablet dated to 1700 BC: The Venus Tablet of Ammisaduqa (see below image). It was found at the famed Library of Nineveh by archaeologist Austen Henry Layard in 1849, and taken to England where it can now be found in the British Museum. This was the same collection which gave us the Epic of Gilgamesh. The Venus Tablet is part of a longer text on Babylonian astrology consisting mostly of omens interpreting celestial phenomena. But it also lists the first and last visible risings of Venus over twenty years (Rawlinson & Smith 1870).
Some historians see the primordial tale of Inana’s descent to the underworld as a precursor to the Judeo-Christian fallen-angel archetype (Jarrell 2013), and Lucifer’s eventual adoption as a name for Satan, as a reinterpretation of Venus’s movements across the heavens (Article “Lucifer” on Jewish Encyclopedia, Vol 8, pg 204). Venus is even mentioned a few times in the Bible, most notably in a pejorative reference to the King of Babylon: “How you have fallen from heaven, morning star, son of the dawn! You have been cast down to the earth, you who once laid low the nations!” (Isaiah 14:12, NIV) We can see here a clear reference to the celestial movements of Inana/Ishtar, which was known to be an important symbol in Babylonian religious traditions. The Jewish author of Isaiah mocks the fall of Babylonian Empire using its own religious imagery.

Fig. 3.77 The Venus Tablet of Ammisaduqa, from Fæ, via Wikimedia (CC BY-SA 3.0)#
Venus from Ancient Greece to 1963#
More than two thousand years ago, Greek scholars established that the Earth was a sphere, and guessed that all the stars were suns, and that suns were giant balls of fire, ours simply being very close by. But as for the other planets in our solar system, it’s not clear when the idea that they might also be actual worlds, as opposed to ‘wandering stars’, first took hold. It’s possible the Pythagoreans believed the planets were terrestrial locations, like the Earth. In one of Aristotle’s critiques of the school, he mocks their ridiculous notions concerning the planets, such that the Earth is just one world among many. But the source material he mocks is lost, and even his own complaints come to us second-hand from Plato. It’s also very possible he’s attributing ideas to the Pythagoreans that they didn’t actually hold. At the very least we know that the concept of the planets as worlds existed, even if only as a thought experiment. In the second century AD, Lucian of Samosata wrote what many consider to be the first work of science fiction, utilising this old Pythagorean heresy for his own narrative purposes. He called his book, A True Story, and it was meant to satirise tall-tales like the Odyssey. (Side note: Venus was the only planet mentioned by Homer in the Iliad, where he called her “Hesperus, the fairest star in the heavens.”)
Lucian’s book was not meant to be taken literally, and it was by no means scientific. Yet still, it was incredibly forward-thinking. It discusses themes like: space travel, aliens, first contact, interplanetary warfare and imperialism, artificial atmospheres, telescopes, robots, and more. It’s an author-insert describing the various fantastical adventures he and his crew have when their ship blows off course during an ill-fated trip across the Atlantic. Amongst a whole series of unfortunate events, they get taken up by a tornado to the Moon, where they bumble into a war between the King of the Moon, Endymion, and Phaethon, the King of the Sun over the colonisation of The Morning Star. While recalling his woes to Lucian, King Endymion calls Venus “empty and uninhabited.” The book was a parody, and meant to be read as a criticism both of popular literature and current events. But it also makes the very intriguing decision to present Venus not as a star, but a terrestrial locale. Was Lucian ahead of his time, or just being as ridiculous as possible (as many contemporaries complained he was wont to do)? Was he part of that scholarly minority hinted at by Aristotle, one who believed “the wandering stars” were worlds, just like Earth? It’s certainly possible.
The widespread understanding of the planets as places like the Earth came to the fore with Copernicus and his heliocentric model in the 16th century. Only then did the paradigm begin to shift, and the general population accept that Earth was part of a collection, a wanderer itself following its own elliptical path around the sun. And after the telescope was invented in the early 1600s scientists began to question in earnest if the other planets had life of their own; whether similar to that on Earth, or wildly different.
The French astronomer Pierre Gassendi (1592-1655) wrote an interesting book called, ‘Are the stars habitable?’ He drew the interesting conclusion that the varying amounts of solar radiation which reached the planets in our system would receive, based on their distance from the sun, would likewise have an affect on the planets and the life therein. Although this is obviously true in a chemical and evolutionary sense, like many scientists of the day, he drew non-sequitur moral inferences. So we have to imagine that the inhabitants of Mercury were much smaller and less perfect than the Venusians and they in turn were inferior to the Earth’s inhabitants.
Bernard de Bovier de Fontenelle (1657-1757) also believed in Venusian inhabitants in his book “Entretiens sur la Pluralité des Monde” published in 1686. It was the first well-founded scientific novel about extraterrestrial worlds. The author spoke about civilisations on Mercury, Venus and Saturn. Fontenelle’s novel was the most popular book of this type until the end of the 18th century. The Dutch astronomer Christian Huygens (1629-1695) extended the ideas of inhabited worlds in the solar system. After observing spots on the surfaces of Jupiter and Mars he came to the conclusion that there were clouds and water there.
So Venus was a kind of cultural touchstone for a long while, representing to most cultures the feminine mystique. But eventually the human race figured out that the stars weren’t gems or candles in a firmament, or mystically imprisoned gods or heroes, but planets and massive balls of gas a long way off. By the 1700s we knew that Venus was the second planet from Sol and that it was similar in size and composition to the Earth, and that it had a very thick atmosphere. This got people’s imagination going in a different fascinating direction. We couldn’t see anything behind the clouds, so there could be anything there. Right away people started thinking about life on Venus.
This interest in life was further fueled by Arthur Eddington, who performed a simple thermal balance calculation for Venus, a calculation you may remember from a previous lecture:
where \(T_{\rm eq}\) (K) is the equilibrium temperature expected on the surface of the planet, \(T_{{\rm eff},*}\) (K) is the effective (blackbody) temperature of the star, \(A_B\) is the Bond albedo, \(R_*\) (m) is the radius of the star, and \(a\) (m) is the semi-major axis of the planet orbiting the star.
For Venus, putting in the values for the Sun and for its orbit, and a Bond albedo of 0.7, that Venus would be expected to have a surface temperature of about 246 K (-27 \(^{\circ}\)C), which is pretty close to Venus’s effective (or brightness) temperature, but too cold for liquid water or Earth-like life. What would he have calculated for the temperature of the Earth? It may have been that Eddington thought Venus’s Bond albedo was lower, say 0.5, in which case surface temperature would be predicted to be around 7 \(^{\circ}\)C. A temperate planet!
The picture changed drastically once the first probe, Mariner 2, got to Venus, and used a microwave radiometer to measure Venus’s surface temperature. Already, there were some hints, from early papers, that the amount of carbon dioxide in Venus’s atmosphere, and the shape of the carbon dioxide spectral lines, very broad, hinted at a very hot surface indeed! But the scientific community, let alone the public, was not so sure until we looked to find out.

Fig. 3.78 Speculation about Life on Venus. From giant extraterrestrial turnips hurled from the Moon onto Venus to defeat the Sun’s warriors with their terrible stench, to the New York Times article about the temperate surface of Venus, to two novels about life on Venus: Asimov’s “Lucky Starr and the Oceans of Venus” and C.S. Lewis’s “Perelandra”.#
Venus Observations#
The Mariner 2 observations suggested the surface of Venus had temperatures around 775 K (about 500 \(^{\circ}\)C). This ended speculation about life on the surface almost entirely, though some speculation about life in the clouds has started up. I’ll briefly discuss this speculation later, in the next lecture.
Since Mariner 2, a whole host of Russian and US probes were sent to Venus, many of which traveled through the clouds and landed on the surface. A great overview of these missions and their results can be found in the two hefty volumes, Venus I and Venus II (Hunten et al. 1983, Bougher et al. 1997).
What I will present here is a “consensus” picture of Venus, its atmosphere, surface and subsurface, based on remote observations, probe observations and some modeling.
Below, you can find a picture of the surface of Venus and a table of what the probes measured of its surface composition.

Fig. 3.79 Surface of Venus. Image from Venera 14, Camera 2 (Russian Academy of Sciences)#
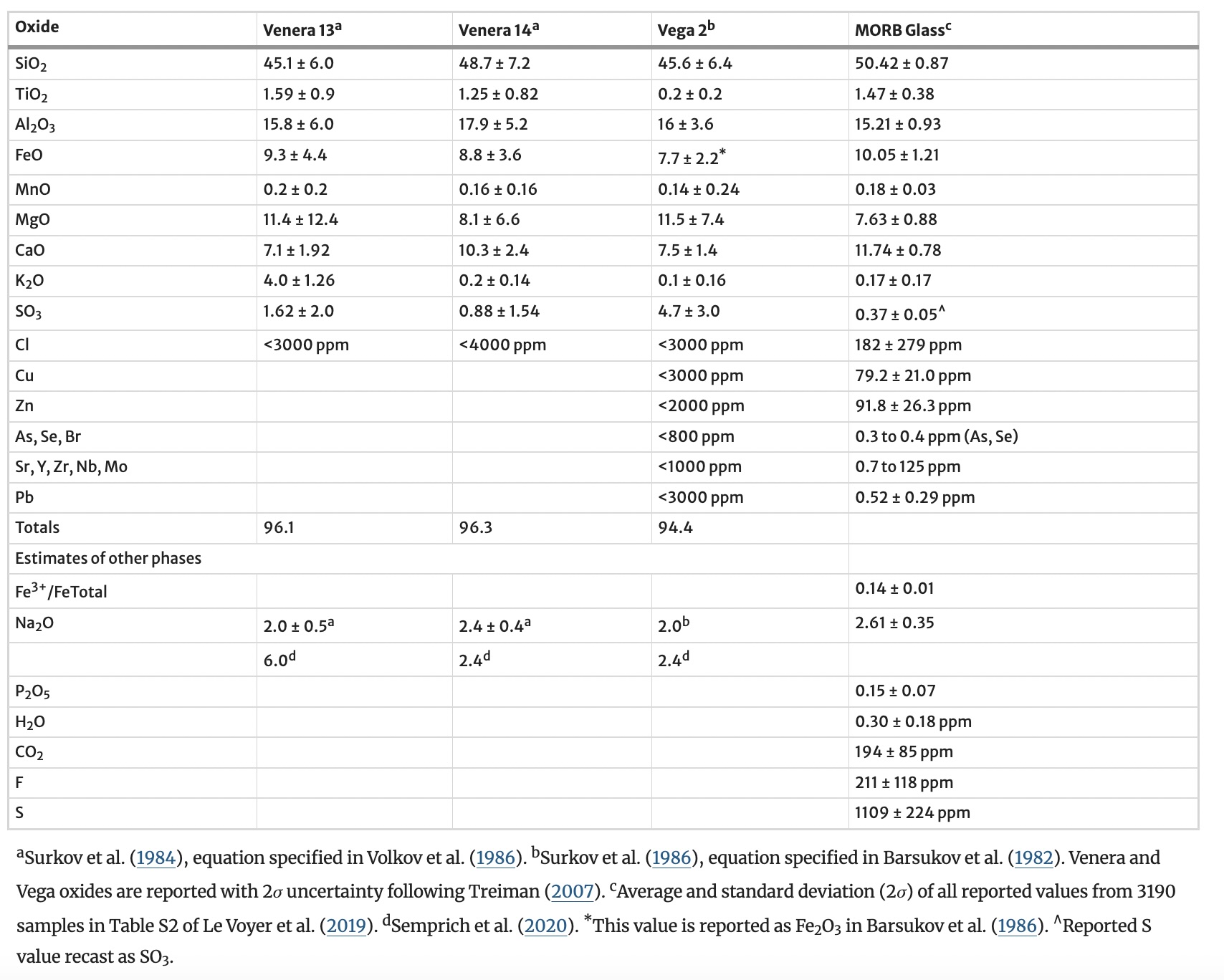
Fig. 3.80 Probe Measurements of the Surface Composition of Venus. Compared to MORB (Mid-Ocean-Ridge Basalt). From Table 2, “X-ray Fluorescence chemical analyses (wt%, 2\(\sigma\)) conducted by the Venera and Vega landers, compared to averaged values for MORB glass”, Gilmore et al. (2023).#
It turns out we can find some consistent mineralogy that fits (most of) the surface data, within reasonable uncertainty, but then we have trouble when we compare this with the gas-phase: the atmosphere in contact with the rock. We don’t have great constraints for either atmosphere or surface, so maybe they are in equilibrium and our data is wrong. Or maybe they are in disequilibrium, and then the data could be correct.
Not shown, but most of the atmosphere is made of CO\(_2\). About 97%. The other 3% (also not shown) is N\(_2\). These aren’t shown because they don’t really do anything interesting. They are constant throughout the atmosphere, until you get very high up. The atmosphere itself is very sulfur-rich, much more than Earth, with 150 ppm SO\(_2\), and 10 ppm OCS. It also has a significant amount (1 ppm) of hydrochloric acid, and a surprising amount of hydrofluoric acid too! Venus is very dry, with only 30 ppm H\(_2\)O vapor in its lower atmosphere. Some of the data is taken more seriously than other data. For example, the CO and OCS is taken very seriously, because different methods keep getting the same answers. But the in-cloud O\(_2\) measurements are not taken seriously at all, because they were only seen by one probe, using one instrument, and not with other probes so far.
The clouds themselves seem to be made of mostly sulfuric acid, though with a fair amount of water, and some other things in there too, like chlorine, iron, and maybe some phosphorus.
Much of the work on Venus is to try to make sense of this data. First, to determine whether the data is reliable, and then to find out what explains its behavior. Future missions, like RocketLab, DAVINCI, and EnVision, are aimed at validating some of these claimed observations, and testing certain hypotheses about how they can be explained. Next, we can overview some of these hypotheses.
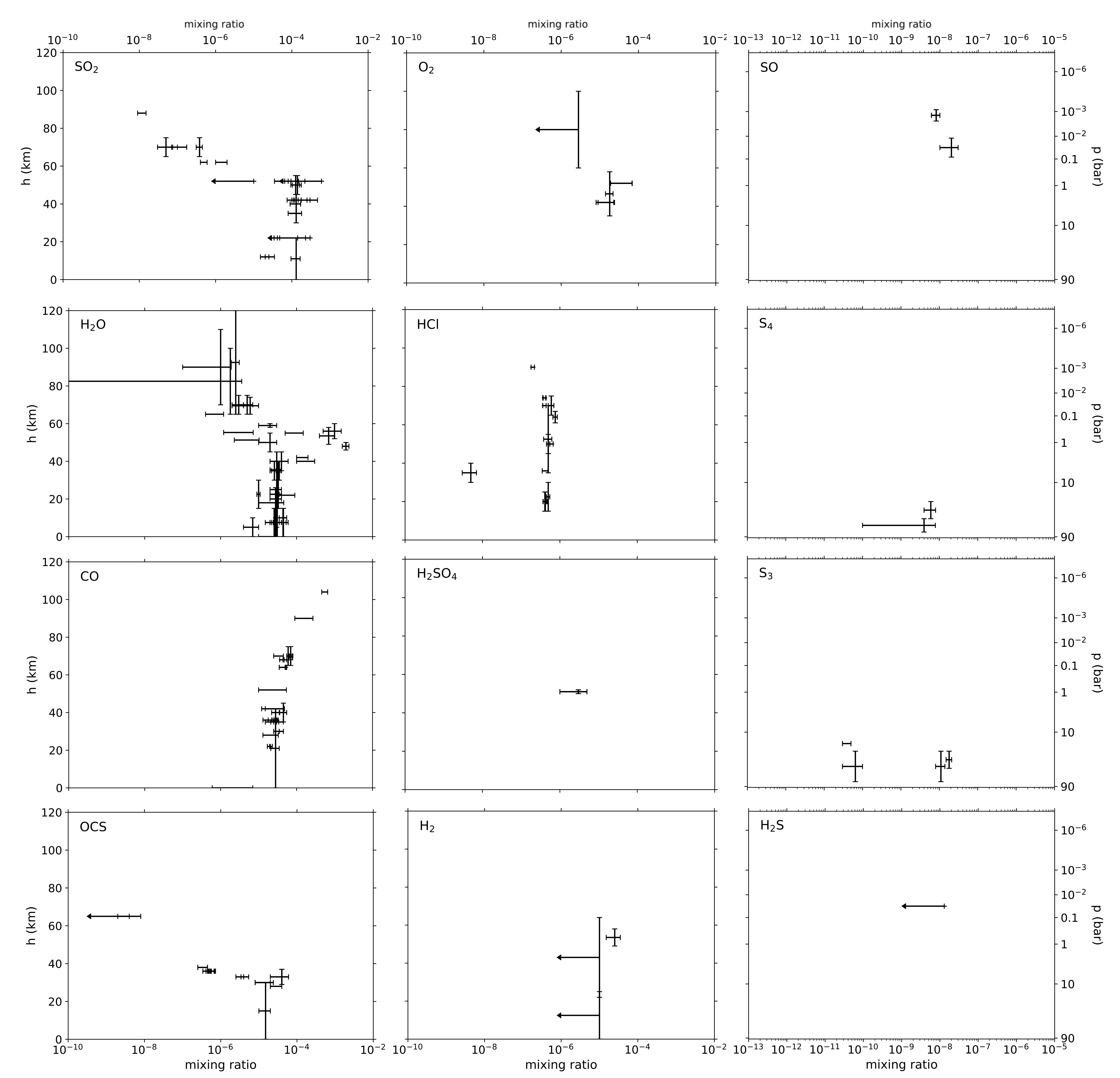
Fig. 3.81 Surface of Venus. Observational constraints on atmospheric composition. From Rimmer et al. (2021).#
Overview of the Atmosphere, Surface and Interior of Venus#
Look at Fig. 3.81. We can see some interesting behaviors with the molecular species with height. There’s also interesting information in horizontal and vertical directions too, but we can only do so much in an hour. And there are already many interesting things to consider.
One of the main things is that jumps out is the depletion of SO\(_2\). It goes away in the clouds. So does H\(_2\)O. The main explanation for this is that the SO\(_2\) and H\(_2\)O react together to form sulfuric acid (H\(_2\)SO\(_4\)). Count up the oxygens, hydrogens, and sulfurs for these three molecules. It doesn’t add up. Given how it doesn’t add up, do you think that the formation of sulfuric acid will oxidize, or reduce, the clouds? In other words, will it take more oxygen out, or add more oxygen in? We will talk more about atmospheric redox in the next lecture, because it is very important for atmospheric evolution.
Another interesting thing we notice is that OCS goes away below the cloud tops. The cloud tops are at ~48 km, and the OCS goes away at ~35 km. Finally, we notice that the CO goes up near the highest part of the atmosphere where data is shown. This can be explained by the breaking of CO\(_2\) by ultraviolet light.
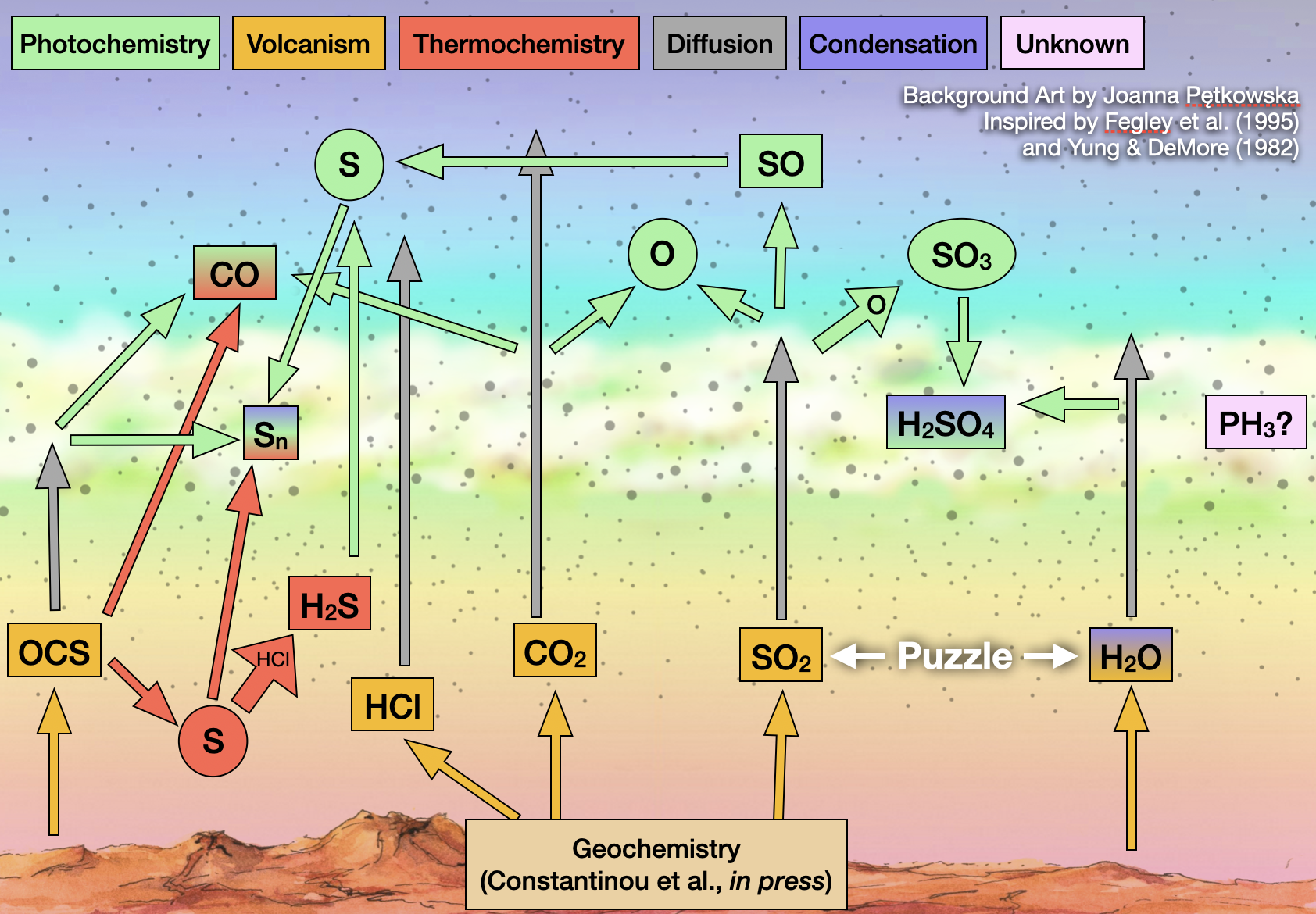
Fig. 3.82 Overview picture of the atmosphere of Venus. Based on figure from Rimmer et al. (2021).#
We already asked whether the surface is in equilibrium with the atmosphere. It can’t be, if all the data is right. What do you think is the biggest contradiction?
What we can do is consider buffers that might represent the global steady-state equilibrium between atmosphere and rocky surface. First, we can find out what the oxygen fugacity (recall from previous lectures) would be for the atmosphere, and match it with an appropriate buffer. This was carried out by Fegley et al. (1997), and can be seen in the figure below.
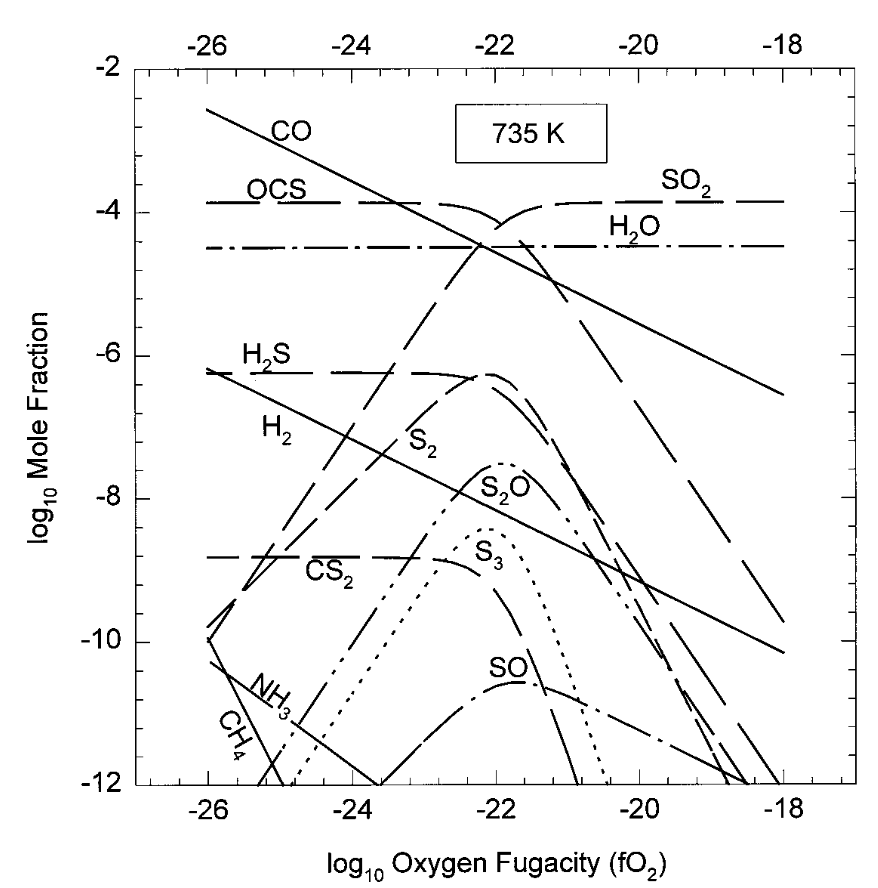
Fig. 3.83 Surface oxidation. On the assumption the lower atmosphere is in equilibrium with the surface. From Fegley et al. (1997).#
We don’t have a lot of data about what’s happening under the ground on Venus, but can make some guesses based on gravimetric data, giving us an idea about bulk interior composition. That, plus some modeling, can give us an idea of what the temperature is beneath Venus’s surface. See the figure below.
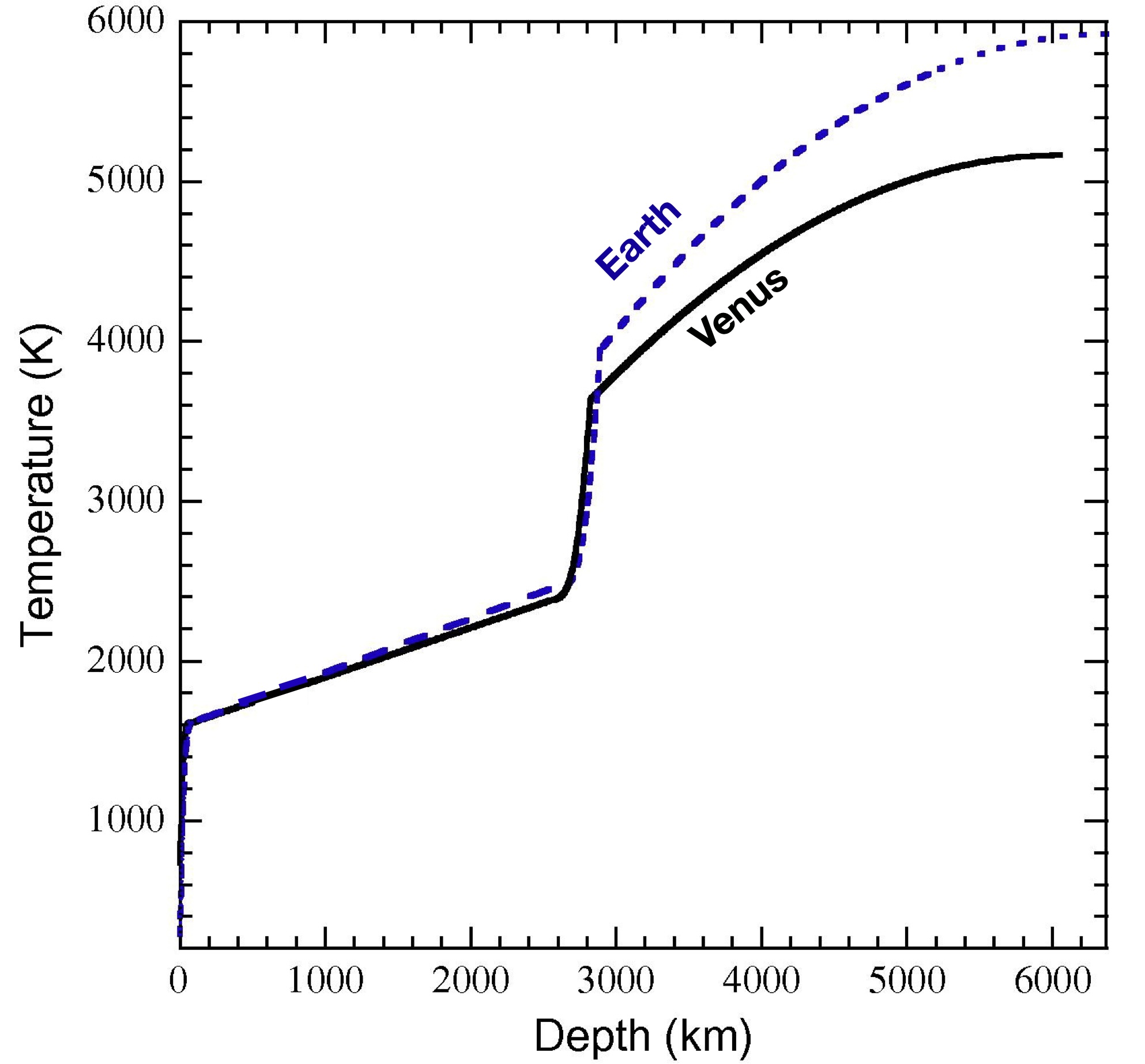
Fig. 3.84 Temperature Inside Venus and Earth. From Fig. 7 of Aitta (2012).#
A Brief Overview of Atmospheric Chemical Kinetics#
You might recall the continuity equation for the atmosphere, describing the change in the number density of a molecular species, \(n_i\) (cm\(^{-3}\)), over time, \(t\) (s):
where \(P_i\) (cm\(^{-3}\) s\(^{-1}\)) is the chemical production of the species, and \(L_i\) (s\(^{-1}\)) is the chemical loss of the species, \(\Phi_{i,z}\) (cm\(^{-2}\) s\(^{-1}\)) is the flux of the chemical species in the vertical, or \(z\) (cm), direction.
There are many powerful implications to this equation, some of which we will return to during C3. But the main implication I want to draw right now involves answering the question: What if there is no production or loss of a particular molecule in the atmosphere? How does that molecule move vertically through an atmosphere, especially an atmosphere in hydrostatic equilibrium and where there is no molecular diffusion (all the molecules move together in the bulk)?
The general expression for the diffusion flux of a minor molecular constituent in an atmosphere is:
where \(K_{zz}\) (cm\(^2\) s\(^{-1}\)) is the Eddy diffusion coefficient, \(P\) (bar) is the atmospheric pressure, and \(T\) (K) is the atmospheric temperature.
The volume mixing ratio of a molecular species in a gas is the number density of the molecular species, \(n_i\), divided by the total number density of the gas, \(n\) (cm\(^{-3}\)). We can call this \(f_i = n_i/n\), a unitless value. Now we replace \(n_i\) with \(nf_i\), and note that, for an ideal gas, \(P = nkT\), where \(k\) is Boltzmann’s constant. This leaves us with:
This means that there will be a flux of the molecular species that has the opposite sign of \(\partial f_i/\partial z\), so diffusion will oppose any gradient in the mixing ratio.
If a molecular species is stable in an atmosphere, it should have a constant mixing ratio with atmospheric height. If it does not, this means that something is making or destroying the molecular species.
The Enduring Mysteries of Venus’s Atmosphere#
List of Mysteries:
Unknown UV absorber: What is it? See Pérez‐Hoyos et al. (2018) to look at newer observations of the absorber (the very first were made in the 1920’s!), and some ideas about what the absorber might be.

Fig. 3.85 UV image of Venus. Processed using ultraviolet (365nm & 283nm) filtered images of Venus taken by Akatsuki on December 23 2016. JAXA/ISAS/DARTS/Kevin M. Gill#
Stability of CO\(_2\): It turns out that CO\(_2\) breaks apart when exposed to ultraviolet light \(\lesssim\) \(200\) nm.
There would be no problem if the reverse reaction (‘M’ is any third body):
were efficient. But it is not. It turns out reactions are slow if the spins of the molecules reacting don’t match. This is also a big problem for CO\(_2\) stability on Mars. But for Mars, there is enough water vapor, compared to the amount of CO\(_2\), which allow for the reactions:
And the H\(_2\) escapes. There is enough water that can be photodissociated in Mars’s atmosphere, compared to CO\(_2\), for this process to stabalize CO\(_2\). If Mars did not have enough water, presumably the CO\(_2\) would all be converted to CO and O\(_2\). Venus does not have enough water for this to work, and yet it still has an atmosphere of 97% CO\(_2\), and surface concentrations of 10 ppm CO. What keeps the CO\(_2\) stable? Researchers have proposed surface processes, and atmospheric chemistry involving chlorine or sulfur. See Yung & DeMore (1982).
Sulfur depletion in the clouds: You can see from the data that SO\(_2\) disappears in the clouds. From the rules about atmospheric mixing, this means that SO\(_2\) must be removed throughout the cloud, and restored again below the cloud. Where does the SO\(_2\) go?
Some of the SO\(_2\) will go to make the sulfuric acid cloud droplets. That makes sense. You can see from the data that there’s 150 ppm SO\(_2\) below the clouds and 30 ppm H\(_2\)O below the clouds. Sulfuric acid is H\(_2\)SO\(_4\), or one molecule of H\(_2\)O, one molecule of SO\(_2\) and one extra oxygen atom. If all the H\(_2\)O goes away, that will only remove about 20% of the SO\(_2\). Where does the rest of the sulfur go?
People have proposed solutions to this problem, including your professor (see Rimmer et al. 2021), but so far, no solution really works.
Sulfur restoration in the middle atmosphere: The SO\(_2\) goes away in the clouds, so it is depleted, from 150 ppm at the bottom of the clouds, altitude 48 km, to \(<1\) ppm at the top of the clouds, altitude roughly 62 km. But then, mysteriously, the SO\(_2\) starts coming back even higher, at \(> 80\) km. Maybe this is due to sulfuric acid droplets getting dredged up very high in the atmosphere, evaporating in a warm spot in the upper atmosphere, a temperature inversion hinted at, and then releasing SO\(_2\) that diffuses down and turns slowly back into sulfuric acid. This at least is the best working hypothesis, proposed by Zhang et al. (2010)
Is the surface in equilibrium with the lower atmosphere? No one knows. Some people think so, some think not.
Loss of OCS below the clouds. Some unknown chemistry needs to be going on to remove OCS and restore CO at 35 km. This mystery was first noticed by Yung et al. (2009).Yung et al. propose some mystery species X to convert OCS to CO, but it’s not clear what this X could even be. Bierson & Zhang (2020) talk about some possibilities.
References#
Aitta, A., 2012. Venus’ internal structure, temperature and core composition. Icarus, 218(2), 967.
Asimov, I., 1987. Lucky Starr and the Oceans of Venus. Del Rey.
Hostetter, C., 1991. Star Trek to Hawa-i’i, San Luis Obispo, California: Diamond Press, p.53
