5.2. Prebiotic chemisty II: Chemical Basis for Life#
Professor: Ziwei Liu (Earth Sciences)
Learning Objectives:
Recognise life’s building blocks and how are they synthesized under different scenarios? (continue)
Understand what homochirality is and how can it be achieved under different scenarios
Direct nucleosidation abiotic nucleotide synthesis (continue)#
Abiotic purines synthesis#
The earliest abiotic synthesis of purine nucleobase was disclosed in 1960 by Orò, who detected adenine when a solution of various concentrations (1.0-11 M) of ammonium cyanide was maintained below 100 °C for several days.
Disadvantages of the HCN polymerization reaciton:
The products are a complex mixture of different nitrogenous bases and HCN polymers, including compounds like azulmin or azulmic acid.
The reaction requires specific conditions to produce adenine and guanine.
It requires a high concentration of HCN, which would have been challenging to achieve under early Earth conditions.
Although the reaction can occur at lower temperatures, the yields are negligible under such conditions.
Abiotic pyrimidines synthesis#
In 1966, Orgel and colleagues proposed cyanoacetylene as a key prebiotic precursor for pyrimidine synthesis. Cytosine was synthesized in a 5% yield by fusing cyanoacetylene with urea in a sealed tube, or by heating potassium cyanate (1.0 M) with cyanoacetylene (0.1 M) at 100 °C for 1 day. Ferris et al. observed similar results, producing cytosine by reacting cyanoacetylene (0.2 M) with cyanate (2.0 M) at 30 °C. Hydrolysis of cytosine yielded uracil.
Direct nucleosidation approaches#
The direct coupling approach for synthesizing ribonucleosides was pioneered by Orgel in the early 1970s. In his experiments, purine nucleosides were formed in low yields with the correct isomer; however, pyrimidine nucleosides were challenging to synthesize due to the kinetic barrier caused by electron delocalization in pyrimidines.
Other groups, including Benner, Carell, Jaber, Zare, and Cronin, enhanced yields and reaction efficiency under prebiotic conditions but did not fully resolve the regioselectivity and stereoselectivity challenges inherent in nucleoside synthesis. While some pyrimidine products were obtained, their yields and purity were much lower than those of purine products, reflecting the inherent difficulties in forming pyrimidine glycosidic bonds under prebiotic conditions.
Moreover, three distinct conditions are required for the synthesis of sugars, purines, and pyrimidines; otherwise, the reaction between formaldehyde and cyanide leads predominantly to the formation of amino acid precursors.
Indirect nucleosidation abiotic nucleotide synthesis#
Direct nucleosidation approaches have had minor successes with purine (adenine) nucleosides, but the production of pyrimidine nucleosides remains unresolved. To address this challenge, Orgel, as a pioneer, mimicked the purine de novo biosynthetic pathway, where the nucleobase is constructed incrementally on the sugar unit. The reaction begins with D-ribose-5-phosphate reacting with cyanamide, resulting in the formation of a ribo-amino-oxazoline intermediate. Subsequent addition of cyanoacetylene to this intermediate yields α-5’-cytidylic acid with a 40% yield, which can then be hydrolyzed to α-cytidine in 10%-20% yield. Notably, α-cytidine can epimerize to the canonical β-cytidine in approximately 5% yield.
In 2009, Sutherland focused on the aminooxazoline intermeidate and systematically investigated its chemistry for the synthesis of pyrimidine nucleotides starting from glyceraldehyde, glycoladehyde, cyanamide, cyanoacetylene and phosphate (Fig. 5.7). Their approach found more acceptance in a prebiotic scenario as it does not start from ribose (previously presumed to be available through the formose reaction) but began with glycolaldehyde and glyceraldehyde, two simpler building blocks that are thought to be more prebiotically plausible.
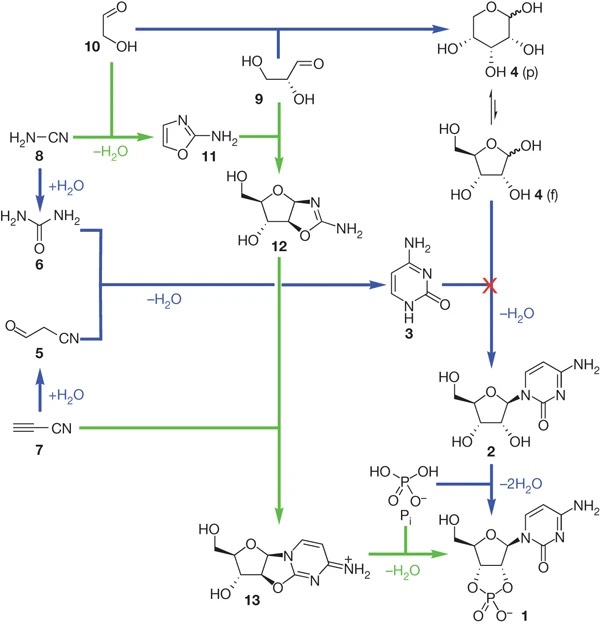
Fig. 5.7 Pyrimidine ribonucleotide assembly options (figure from Sutherland, et al., (2009) Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions).#
It is worth pointing out that, in this synthesis, all the starting materials are also precursors of amino acids under similar conditions. Furthermore, these starting materials are used not only in the synthesis of amino acids but also in the synthesis of phospholipids (Fig. 5.8).
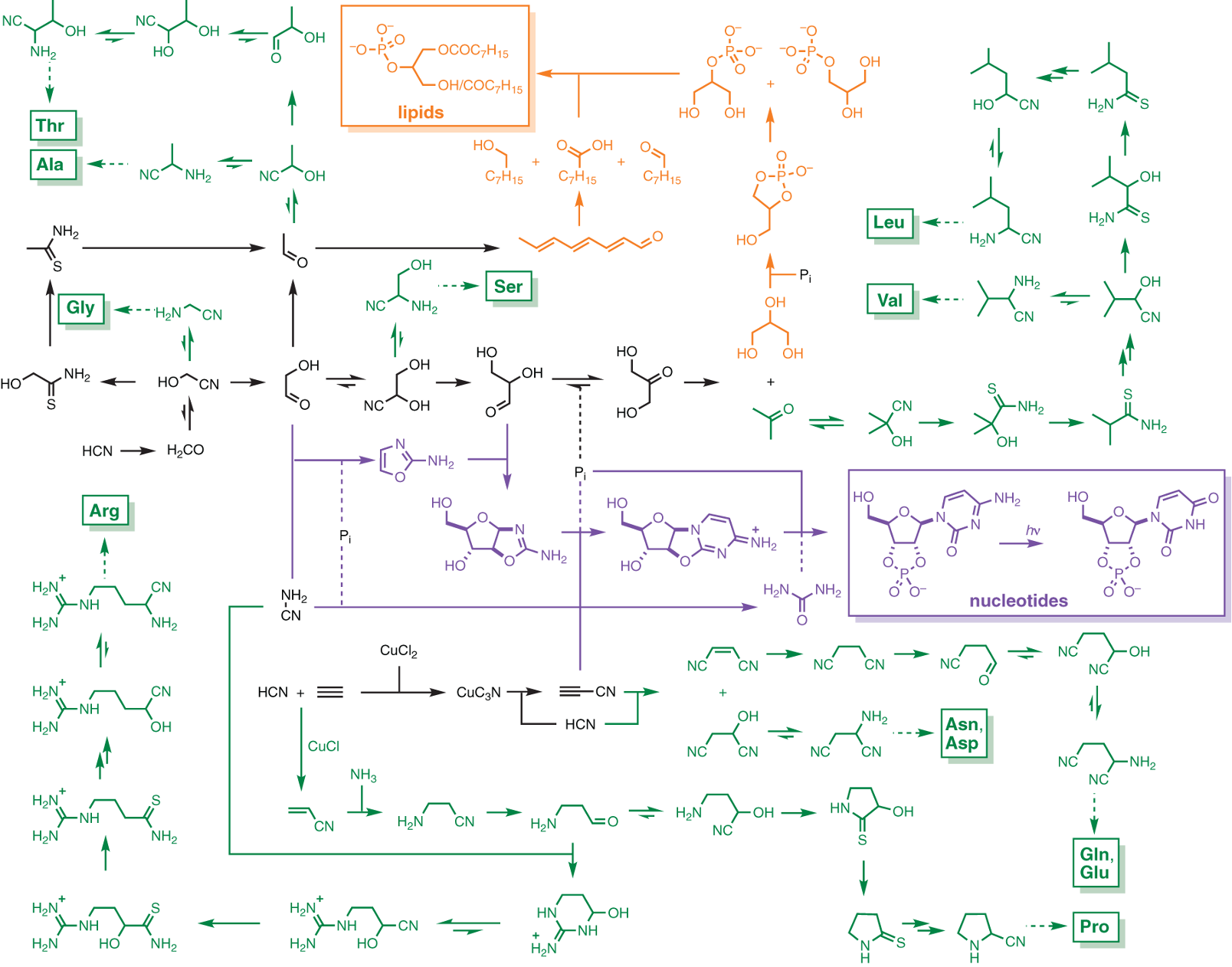
Fig. 5.8 Cyanosulfidic protometabolic reaction network leading from HCN and derivatives thereof to pyrimidine ribonucleotides, and amino acid and lipid precursors (figure from Wu, et al., (2019) Provisioning the origin and early evolution of life).#
Homochirality#
Definition#
Homochirality refers to the uniformity of chirality (handedness) in biological molecules. Chirality is a property of molecules that can exist in two non-superimposable mirror-image forms, known as enantiomers. For instance:
Amino acids (the building blocks of proteins) predominantly exhibit the L configuration (left-handedness).
Sugars (key components of nucleic acids like DNA and RNA) predominantly exhibit the D configuration (right-handedness).
Importance of homochirality#
Homochirality is a fundamental characteristic of all known life forms and plays a crucial role in the structure and function of biomolecules. Its significance lies in the following aspects:
Molecular Recognition and Functionality: Biological processes such as enzymatic catalysis, protein folding rely on highly specific molecular interactions. Homochirality ensures that molecules fit together properly, much like a lock and key. A mix of chiralities will disrupt these processes.
Structural Integrity: The uniform chirality of amino acids and sugars contributes to the stable, repeatable structures of proteins and nucleic acids. For example, the double helix of DNA relies on consistent sugar chirality for its shape.
The potential driving force for the origin of homochirality#
Parity-Violating Energy Difference
Parity violation refers to the phenomenon where the laws of physics are not symmetric under a mirror reflection. This subtle asymmetry could influence the preference for one enantiomer over its mirror image.
Circularly Polarized Light (CPL)
Circularly polarized light acts as a chiral agent and interacts with molecular chirality through circular dichroism. Chiral molecules exhibit optical activity by selectively absorbing CPL, which can induce or amplify chirality in certain chemical reactions.
Frank Model
In 1953, Frank proposed a “simple and sufficient life model” to explain the emergence of homochirality. He described an autocatalytic system in which a chiral compound acts as a catalyst to enhance its own production while simultaneously suppressing the production of its mirror image. This mechanism can amplify a tiny initial imbalance in chirality, leading to the dominance of one enantiomer.
Soai Reaction
The first experimental proof of the Frank model was reported in 1995 through the autocatalytic alkylation of achiral pyrimidyl aldehydes with dialkylzincs in toluene. In this reaction, the chiral alcohol product serves as an asymmetric catalyst for its own production, providing a practical demonstration of homochirality amplification.
Conglomerate Crystallization
Chiral crystals can form in three primary types:
Racemates: Composed of equal proportions of both enantiomers in an orderly arrangement within the crystal lattice (~90% of chiral crystals).
Solid Solutions: Containing both enantiomers in a disordered arrangement (rare).
Conglomerates: Containing only one enantiomer in individual crystals (~10% of chiral crystals).
Conglomerate crystallization can naturally enrich one enantiomer, providing a potential pathway to homochirality.
Chiral-Induced Spin Selectivity (CISS) Effect
The CISS effect is a quantum mechanical phenomenon where chiral molecular structures preferentially interact with electrons of a specific spin orientation. Chiral molecules create a helical electric field when electrons move through them, coupling the electron’s spin and momentum. This results in spin-polarized electron transport, which may influence chirality selection in chemical and biological systems.
Chiral information propagation#
Under metabolism-first scenario
In 1848, Louis Pasteur discovered molecular chirality while studying the optical activity of sodium ammonium tartrate crystals which can form enantiomorphous conglomerates. Tartrate is a precursor to malate (Fig. 5.9). However, when malate (the blue spot) enters the Krebs cycle, it either undergoes dehydration to form fumarate (clockwise) or oxidation to produce oxaloacetate (anticlockwise), both of which are achiral compounds. Similarly, the homochirality established at isocitrate (the red spot) also meets the same fate. Furthermore, there are 19 chiral proteinogenic amino acids, each with its own idiosyncratic synthesis pathway. While it is possible to induce enantiomeric excess in a few amino acids, there is currently no experimental evidence to support this.
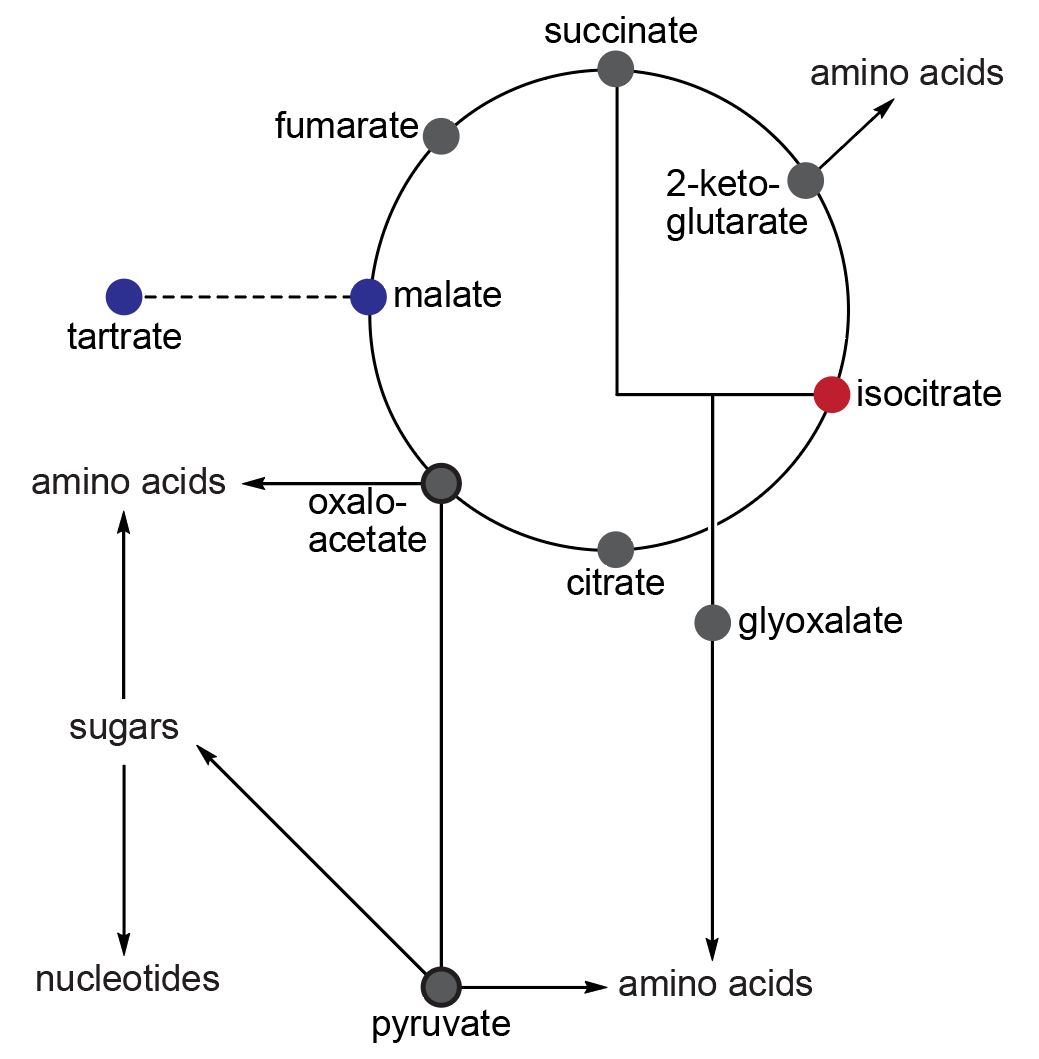
Fig. 5.9 Core metabolism involves two chiral entities (L-malate and D-isocitrate, shown in blue and red respectively) flanked by achiral intermediates (shown in gray). (figure reproduced from Ozturk, et al., (2023) The central dogma of biological homochirality: How does chiral information propagate in a prebiotic network? ).#
Under material-first scenario (Fig. 5.10)
D-RAO as a Precursor: In cyanosulfidic chemistry, D-ribose-aminooxazoline (D-RAO) serves as a common precursor for deoxyribonucleotides and ribonucleotides.
Chirality Selection: Due to its low solubility (compared to arabinose-, xylose-, and lyxose-aminooxazolines) and the CISS effect on spin-polarized magnetic surfaces, enantiomer-pure D-RAO crystals can form.
Propagation to RNA: These crystals yield enantiomer-pure nucleosides, leading to enantiomer-pure RNA.
L-Amino Acids and tRNA Analogs: L-amino acids selectively attach to tRNA analogs composed of D-ribonucleotides, enabling a homochiral genetic molecule.
Peptide Synthesis: Homochiral genetic molecules synthesize peptides enriched with L-amino acids from racemic amino acid mixtures.
Chiral Propagation: Homochiral peptides transmit chiral information to metabolites via enantioselective catalysis, propagating homochirality throughout the prebiotic network.
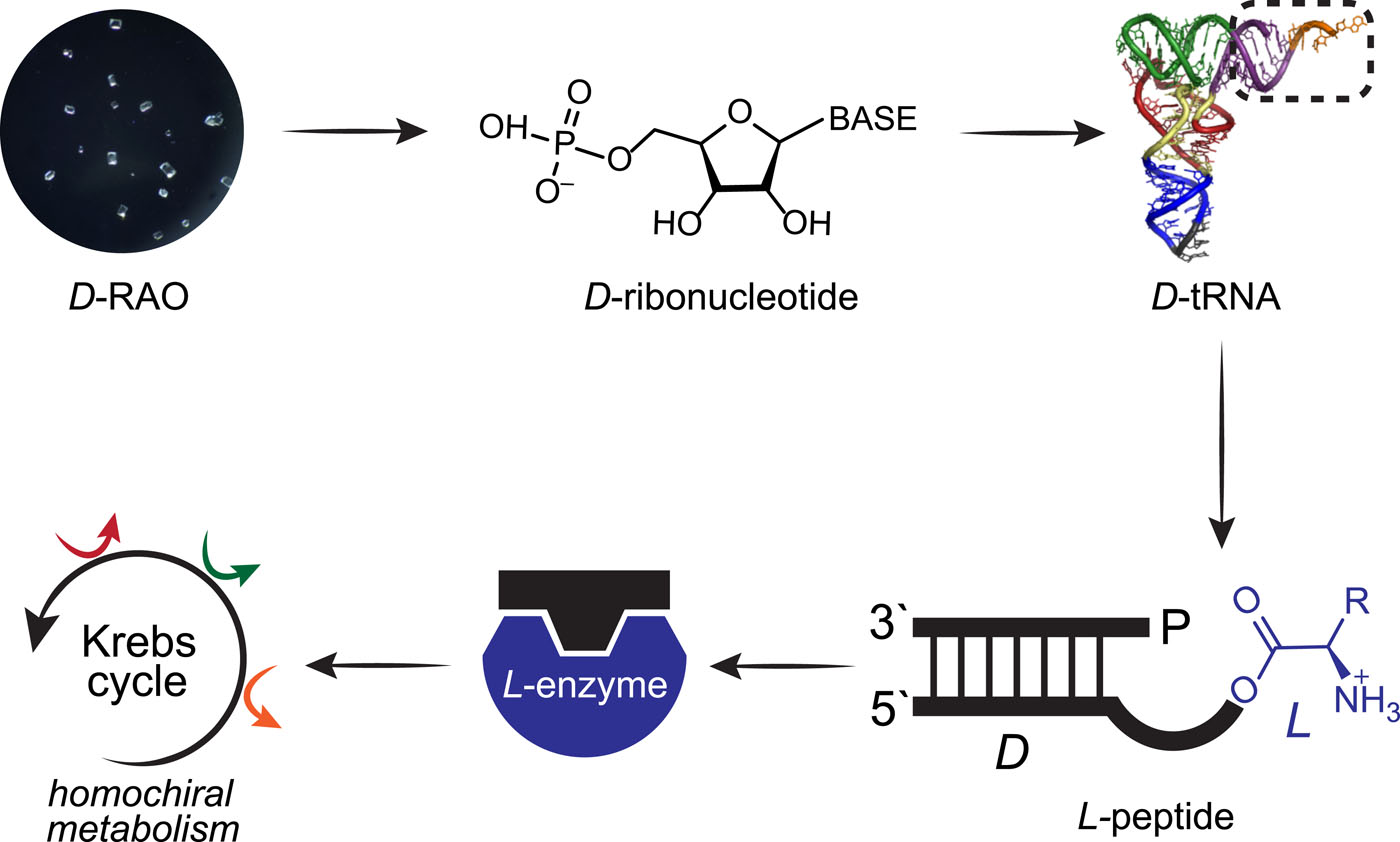
Fig. 5.10 Propagation of homochirality from a common ribonucleotide precursor, RAO, to the entire prebiotic network is illustrated. D-RAO can be transformed into D-ribonucleotides, forming homochiral RNA. L-peptides can be synthesized through the stereoselective binding of L-amino acids to tRNA analogs composed of D-ribonucleotides. This crucial link enables the transfer of homochirality from nucleic acids to peptides. Then L-peptides form L-enzymes, and by enantioselective catalysis a homochiral metabolism can be produced. (figure from Ozturk, et al., (2023) The central dogma of biological homochirality: How does chiral information propagate in a prebiotic network? ).#
Reference#
Wächtershäuser G., (1992) Groundworks for an evolutionary biochemistry: The iron-sulphur world
Sutherland J., (2016) The origin of life - Out of the Blue
Yadav M., Kumar R., Krishnamurthy R., (2020) Chemistry of Abiotic Nucleotide Synthesis
Liu, (2021) Prebiotic photoredox synthesis from carbon dioxide and sulfite
Ozturk, (2024) A New Spin on the Origin of Biological Homochirality
Xu J. et al., (2020) Selective prebiotic formation of RNA pyrimidine and DNA purine nucleosides
Xu J. et al., (2021) Prebiotic photochemical coproduction of purine ribo-and deoxyribonucleosides
