5.1. Prebiotic chemisty I: Chemical Basis for Life#
Professor: Ziwei Liu (Earth Sciences)
Learning Objectives:
Understand what prebiotic chemistry is and why it is important for understanding the origin of life
Know how can we break down modern life into subsystems, and what is the origin-of-life scenario based on these subsystems
Recognise life’s building blocks and understand how are they synthesized under different scenarios
Prebiotic chemistry: Bridging the gap between planet formation and biological evolution#
What is prebiotic chemistry?#
Earth formed 4.54 billion years ago, and life is believed to have originated around 3.5 billion years ago. The gap between these events is vast and marked by significant chemical evolution. Prebiotic chemistry refers to the chemical reactions that occurred during this time, paving the way for life. It seeks to answer the profound question: how did non-living molecules evolve into living systems? Chemistry play a central role in unraveling these mysteries.
To explore the origin of life, two main strategies are employed:
Geochemistry up (“bottom-up”): Scientists study early planetary environments, examining rocks, minerals, and atmospheres to reconstruct plausible chemical reactions that led to life.
Biology down (“top-down”): By analyzing current biology, researchers infer the molecules, pathways, and structures present in ancient life, tracing back to the Last Universal Common Ancestor.
What is the goal of prebiotic chemistry?#
The goal is to map the journey from simple molecules to protocells (Fig. 5.2). This involves identifying:
Key molecules and their roles.
Chemical pathways that could produce them.
Processes leading to increasingly complex structures like polymers and cellular membranes.
Ultimately, prebiotic chemistry connects the origin of small molecules to the emergence of LUCA.
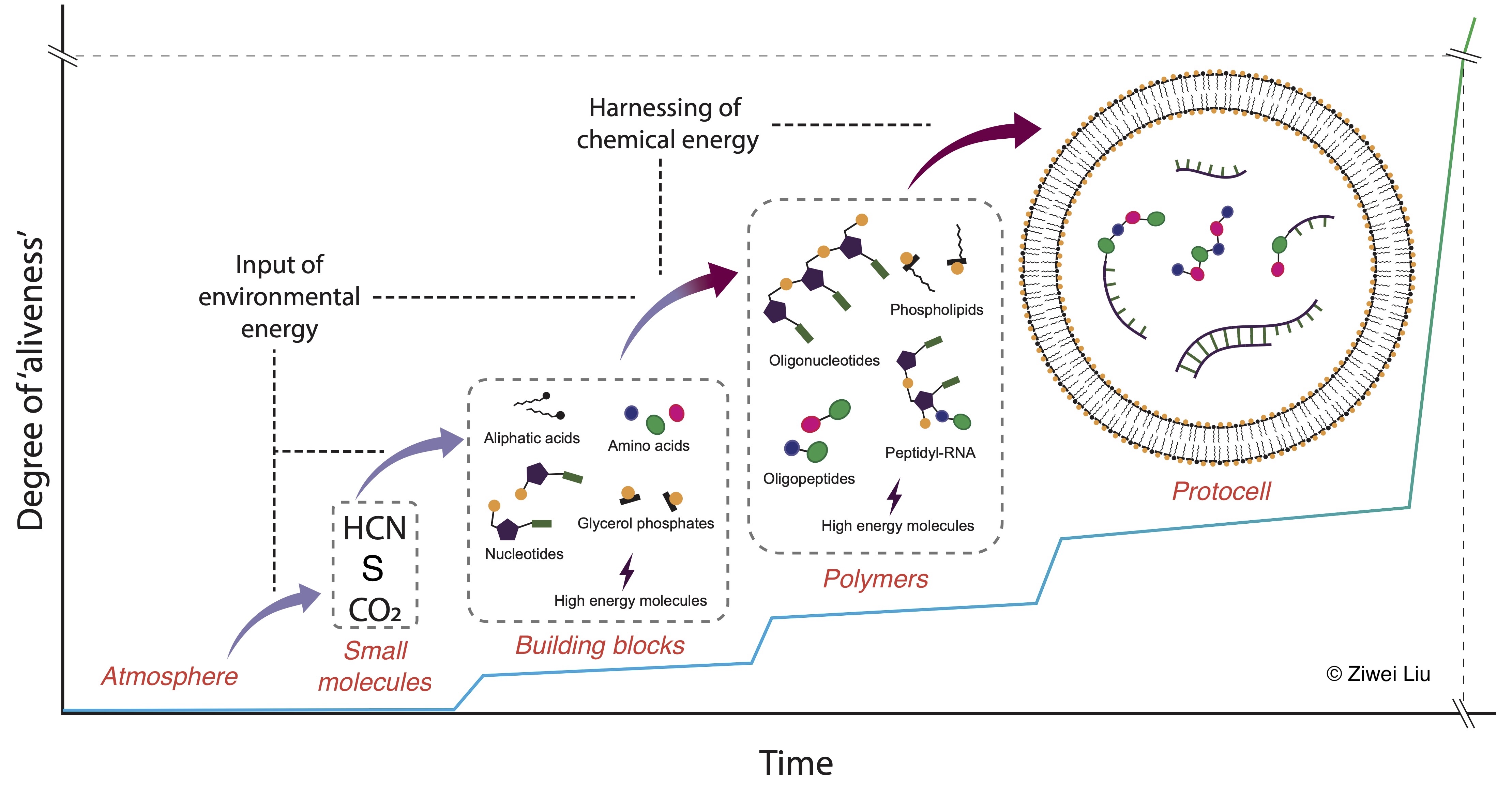
Fig. 5.2 Prebiotic chemistry: the chemical evolution of molecules from the original atmosphere to protocells. More information: https://ziwei-personal-git.github.io.#
System Chemistry: Living systems and their subsystems#
Systems chemistry is an area of chemistry that seeks insight into complex networks of interacting molecules and their system-level properties. These properties emerge through the collective behavior of the system’s components and cannot be attributed to the individual components acting in isolation. The way in which specific interactions between the components propagate through the system dictates these emergent properties.
Living organisms can be broken down into four key subsystems (Fig. 5.3):
Informational subsystems: RNA and DNA, which store and transmit genetic information.
Metabolic subsystems: Networks of chemical reactions essential for energy conversion and biosynthesis.
Catalytic subsystems: Proteins that act as enzymes to accelerate reactions.
Compartment-forming subsystems: Membranes and phospholipids that create distinct cellular boundaries.
These subsystems interact to sustain life, but their origins present a challenge: how did they evolve from non-living chemistry?
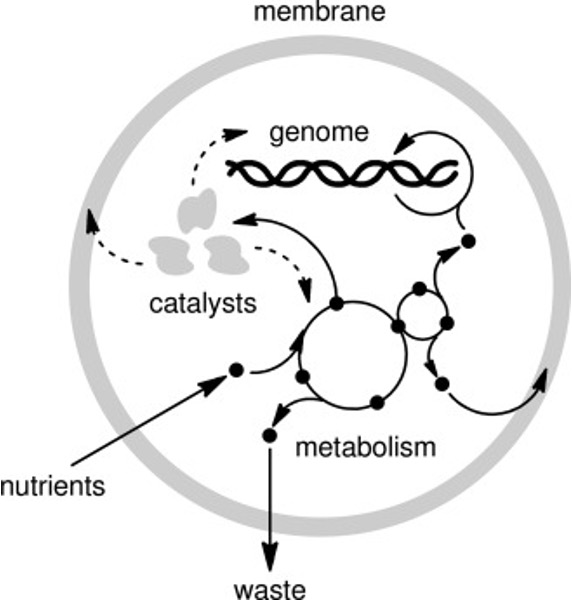
Fig. 5.3 A minimal cell with emphasis on its subsystems (figure from Sutherland J., (2016) The origin of life - Out of the Blue).#
The chicken and egg problem#
Nucleic acids (DNA and RNA) are essential for storing genetic information and directing protein synthesis.
However, proteins are needed to synthesize nucleic acids.
One major question is: which came first, nucleic acids or proteins? This creates a paradox often called the “chicken-and-egg problem” in the origin of life.
RNA world hypothesis#
Historical Milestones:
1967: Carl Woese, Francis Crick, and Leslie Orgel proposed that RNA might act as both a genetic material and a catalyst.
1982: Thomas Cech discovered that RNA could catalyze reactions, such as self-splicing, proving its dual functionality.
The RNA world hypothesis suggests that early life was based on RNA, a molecule capable of both storing genetic information and catalyzing chemical reactions. Evidence for this comes from the discovery of ribozymes, RNA molecules with catalytic abilities. These findings suggest RNA could have performed many functions now carried out by proteins and DNA.
However, the RNA world hypothesis raises new questions:
How did RNA molecules originate from prebiotic chemistry?
How did life transition from an RNA-based system to the DNA-RNA-protein system we see today?
RNA-peptide world hypothesis#
Recent research suggests that life may not have started with an RNA world alone. Instead, it could have originated in an RNA-peptide world, where RNA and small peptides (short chains of amino acids) co-evolved. Peptides might have stabilized RNA structures or enhanced their catalytic functions, creating a mutually beneficial system.
Why is this idea important? The RNA-peptide world hypothesis helps address some limitations of the RNA world model. It suggests that from the very beginning, life might have relied on collaboration between molecules, setting the stage for the complex interplay between RNA, proteins, and DNA that defines life today.
Metabolism first hypothesis#
The Metabolism First Hypothesis proposes that life originated from self-sustaining chemical reactions (metabolic networks) rather than from information-carrying molecules like RNA or DNA. According to this idea, primitive metabolic cycles may have formed in the early Earth’s environment, providing the energy and chemical precursors necessary for more complex biological molecules to emerge.
Chicken and egg problem:
Metabolic networks rely on enzymes to catalyze chemical reactions. Enzymes, in turn, are proteins, which are synthesized through complex biochemical processes. However, this raises a fundamental question: How were proteins initially formed in the absence of enzymes?
The debate between autotroph and heterotroph origins of life#
In 1992, Wächtershäuser stated: “The biosphere is deeply divided between autotrophic and heterotrophic ways of life. This gives biology its primary problem: Was the first organism an autotroph, capable of synthesizing all its carbon constituents from carbon dioxide (or other C1-units) or was it a heterotroph, dependent on taking up organic carbon compounds as food.” This statement highlights the central debate about the origin of life:
Did life begin with autotrophs, organisms that could synthesize their own organic molecules from inorganic substances (“metabolism-first”)?
Or did it start with heterotrophs, organisms that relied on pre-existing organic molecules (“material-first”)?
Autotroph origin of life#
The autotroph-first hypothesis suggests that the first life synthesized their own organic molecules using inorganic compounds and energy from their surroundings.
Key point:
Hydrothermal vents as cradles of life: Early life may have originated near hydrothermal vents, where abundant inorganic molecules like hydrogen, carbon dioxide, and sulfur compounds provided raw materials.
Primitive metabolic pathways: Pathways such as the reductive acetyl-CoA pathway or the reverse Krebs cycle could have facilitated the synthesis of essential organic compounds.
Energy from natural gradients: Hydrothermal vents create natural proton gradients, which might have driven energy production via chemiosmosis.
Challenges:
Complexity of Autotrophic Pathways: Many autotrophic processes require sophisticated enzymes and cofactors, which may not have been available in prebiotic conditions.
Abiotic Formation of Complex Molecules: Demonstrating the abiotic synthesis of key molecules, such as nucleosides, from inorganic precursors remains a significant experimental hurdle.
Heterotroph origin of life#
The heterotroph-first hypothesis suggests that the earliest life forms relied on pre-existing organic molecules in their environment for energy and growth.
Key Points:
Primordial soup hypothesis: Early Earth may have been rich in a “primordial soup” of organic molecules, generated by natural processes like lightning (as demonstrated in the Miller-Urey experiment) or delivered via meteorites.
High energy molecules as a simple energy source: Primitive life forms could have relied on readily available organic compounds to meet their energy needs.
Challenges:
Limited Supply of Organic Molecules: The availability of prebiotic organic molecules may have been insufficient or short-lived, raising questions about how early heterotrophs sustained themselves.
Transition to Autotrophy: The model partially explains how early life transitioned from relying on external organic molecules to synthesizing its own (autotrophic processes; see Photoreduction of carbon dixoide).
Carbon dioxide fixation#
Biology carbon dioxide fixation pathways#
Inorganic carbon enters the biosphere through carbon fixation, a process essential for primary production of organic compounds. Autotrophic carbon fixation converts inorganic carbon into biomass, forming the foundation of the biosphere’s carbon cycle. Currently, six natural autotrophic carbon-fixation pathways are well-described and widely accepted (Table Table 5.1).
Pathway |
Energy Source |
Input |
Output |
Reductants |
Key Enzymes |
|---|---|---|---|---|---|
Calvin-Benson |
Light |
3 CO\(\sf _2\), 9 ATP, 6 NAD(P)H |
Glyceraldehyde-3-phosphate |
NAD(P)H |
RuBisCO |
DC/HB |
Hydrogen and Sulfur |
1 CO\(\sf _2\), 1 HCO\(\sf _3^-\), 3 ATP, 4 NAD(P)H |
Acetyl-CoA |
NAD(P)H and ferrodoxin |
Pyruvate synthase, PEP carboxylase |
3-HP |
Light |
3 HCO\(\sf _3^-\), 5 ATP, 5 NAD(P)H |
Pyruvate |
NAD(P)H |
Acetyl-CoA carboxylase, Propionyl-CoA carboxylase |
HP/HB |
Hydrogen and Sulfur |
2 CO\(\sf _2\), 4 ATP, 4 NAD(P)H |
Acetyl-CoA |
NAD(P)H |
Acetyl-CoA-Propionyl-CoA carboxylase |
rTCA |
Light and Sulfur |
2 CO\(\sf _2\), 2 ATP, 4 NAD(P)H |
Pyruvate |
NAD(P)H and ferrodoxin |
2-Oxoglutarate synthase, Isocitrate dehydrogenase |
Wood-Ljungdahl |
Hydrogen |
2 CO\(\sf _2\), 1 ATP, 4 NAD(P)H |
Acetyl-CoA |
Ferrodoxin |
NAD-independent formate dehydrogenase, Acetyl-CoA synthase-CO dehydrogenase |
Abiotic carbon dioxide fixation pathways#
Abiotic carbon dioxide fixation in prebiotic chemistry can occur through various chemical pathways, often involving the reduction of carbon dioxide (i.e., the addition of electrons or hydrogen) to form simple organic compounds, such as aldehyde, carboxylic acid. These reactions may have been driven by natural energy sources like ultraviolet radiation, lightning, volcanic activity, or hydrothermal vent systems, which provided the necessary conditions for these reactions to occur.
Plasma electrochemical reduction (Miller-Urey type reaction)#
Lightning strikes convert into formic acid and ammonium, which are crucial precursors for biomolecules. These compounds play foundational roles in the synthesis of amino acids and nucleotides, supporting the emergence of life.
The study highlights gas-liquid-solid interfaces (air-water-ground) as critical zones for enhanced chemical activity. These interfaces produce higher yields of reactive compounds compared to reactions occurring in gas-phase-only systems.
Minerals in early Earth’s environment, such as sulfides and carbonates, significantly influence reaction pathways. They increase the efficiency of nitrogen fixation and stabilize reaction products, ensuring a steady supply of necessary compounds.
Spark discharge experiments demonstrate that cloud-to-ground lightning could produce substantial amounts of nitrate, nitrite, and ammonium, essential for prebiotic chemistry and the development of life.
Hydrothermal CO2 reduction#
Starting with either CO2 or sodium bicarbonate, in the presence of different metals and under various conditions of pH, temperature, pressure, and reaction time.
The products are carboxylic acids, methane, phenol, and liquid fuels.
Photoreduction of carbon dioxide#
The photo reduction of CO2 using an aqueous solution of bicarbonate and sulfite under UV irradiation. Converting CO2 into various organic compounds (formate, hydroxymethanesulfonate, methanol, glycolate, acetate, tartronate, malonate, rac- and meso- tartrate, and hydrogen gas)(Fig. 5.4).
Furthermore the photoreduction of glycolate converted it into citrate, malate, and succinate demonstrating the generation of key metabolites relevant to central carbon metabolism.
The conversion yield of CO2 to organic compounds is up to 70%.
Giving an connections between environmental chemistry and the development of metabolism: nascent biology initially relied on the building blocks produced by environmental chemistry (heterotroph scenario), but the depletion of building blockes created evolutionary pressure to synthesize them from other available sources (carboxylates produced in this chemistry). With time, the supply of carboxylates would also dwindle and biology would have to evolve to make do with simpler, only remaining abundant carbon source, namely CO2.
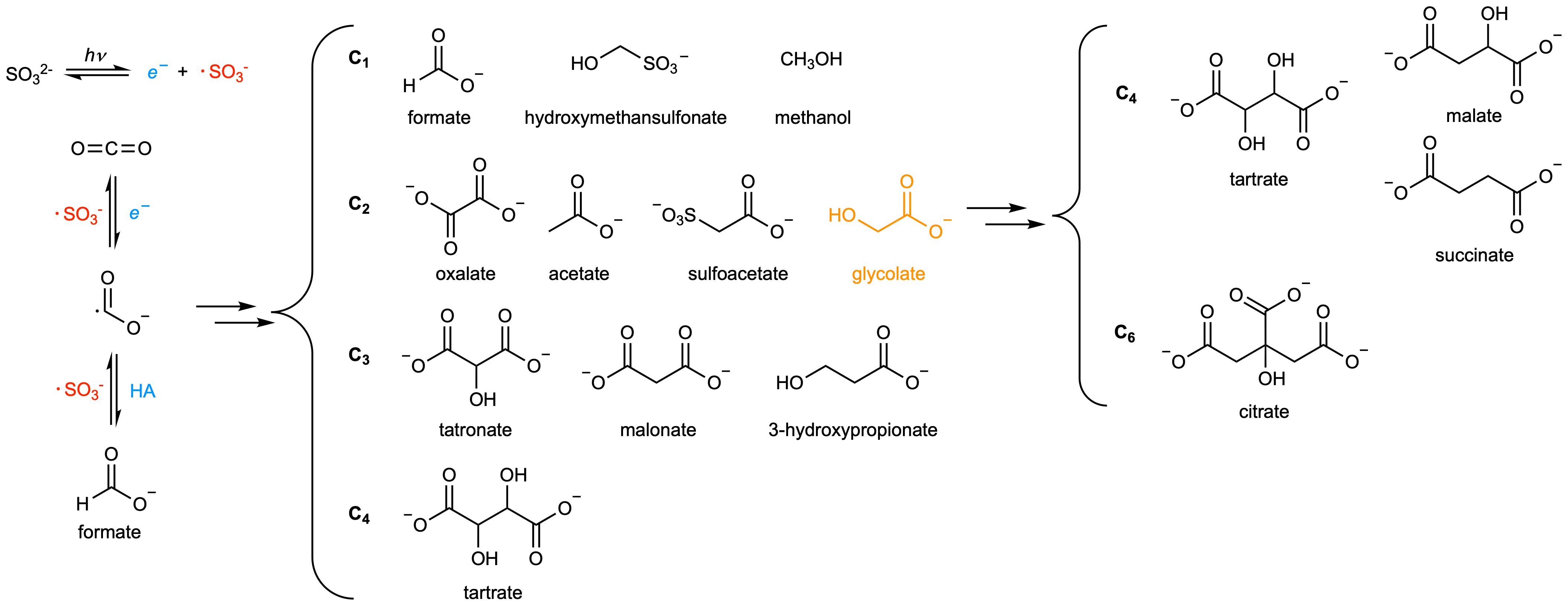
Fig. 5.4 Photoreduction of bicarbonate in the presence of bisulfite (figure from Liu, et al., (2021) Prebiotic photoredox synthesis from carbon dioxide and sulfite).#
Prebiotic building blocks and the synthesis pathway#
Prebiotic building blocks are the basic chemical components and molecules that may have contributed to the origin of life on Earth, long before biological processes and organisms emerged. These molecules likely formed naturally under prebiotic conditions on early Earth or in extraterrestrial environments like meteorites or interstellar clouds. They are essential for the formation of more complex molecules that eventually lead to life, such as nucleic acids, proteins, and lipids.
Amino acids#
Amino acids are the building blocks of proteins and enzymes, which are crucial for biological functions such as catalysis, structure, and signaling (Fig. 5.5).
Structure of Amino Acids#
Each amino acid consists of three functional groups:
A carboxyl group (-COOH).
An amino group (-NH2).
A variable side chain (R group) that determines the properties of the amino acid.
If the carboxyl group and amino group are attached to the same carbon atom, the molecule is classified as an α-amino acid.
Chirality of Amino Acids#
Most α-amino acids are chiral, meaning they exist in two mirror-image forms: L- (left-handed) and D- (right-handed) enantiomers. In biological systems, L-amino acids dominate due to their incorporation into proteins.
Variety of Amino Acids#
There are numerous amino acids in nature, each differing by its side chain. Of these, only 22 proteinogenic amino acids are directly encoded by the genetic code and used to construct proteins in living organisms.
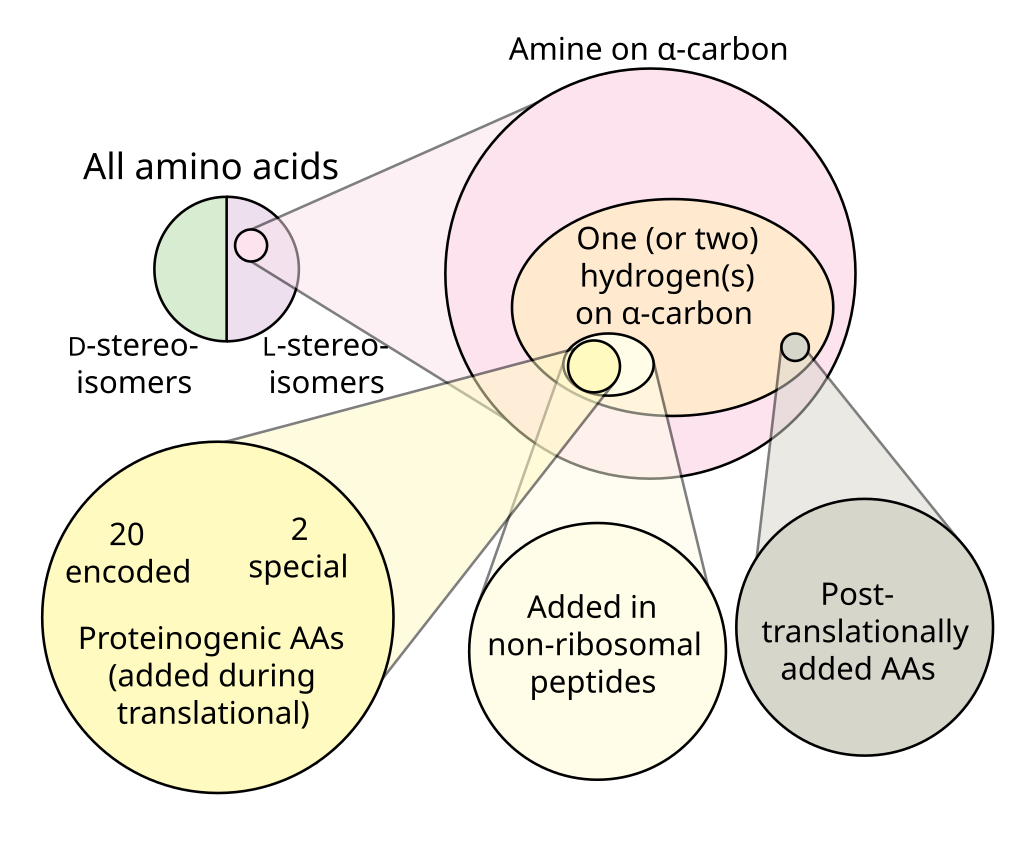
Fig. 5.5 Venn diagram showing that the 22 proteinogenic amino acids are a small fraction of all amino acids (figure from Wikipedia).#
Biologic synthesis of amino acids#
Different amino acids have distinct synthesis pathways.
Glutamine and glutamate are major sources of amino groups for the synthesis of other amino acids.
Transamination is a biochemical process in which an amino group (-NH₂) is transferred from an amino acid to an α-keto acid, producing a new amino acid and a new α-keto acid. This reaction is catalyzed by aminotransferases and requires pyridoxal phosphate (PLP) as a coenzyme.
Reductive amination is a chemical reaction in which a carbonyl group (C=O), typically from an aldehyde or ketone, is converted into an amine (C-NH2) through the addition of an amino group (-NH2) and subsequent reduction, often catalyzed by specific enzymes.
The synthesis of glutamate relies on the reductive amination of α-ketoglutarate (from the TCA cycle) by glutamate dehydrogenase.
Glutamine is synthesized from glutamate by glutamine synthetase in a separate ATP-dependent reaction.
Abiotic synthesis of amino acids#
Abiotic transamination or reductive amination: Uses metal catalysts (e.g., iron or nickel) to replace enzymes in facilitating these reactions under prebiotic conditions.
Strecker synthesis: The Strecker amino acid synthesis is a chemical reaction that produces α-amino acids from an aldehyde or ketone, ammonia (NH3) (or an amine), and hydrogen cyanide (HCN). Notable examples include the Miller-Urey experiment and the HCN homologation reaction, both of which demonstrate the formation of amino acids under prebiotic conditions.
Nucleotides#
Nucleotides are the fundamental building blocks of nucleic acids (DNA and RNA), which are essential for storing and transmitting genetic information within cells. They also play vital roles in various cellular processes beyond their role in nucleic acids, such as energy transfer and cell signalling.
Structure of nucleotides#
A nucleotide consists of three key components:
A Nitrogenous Base: Purines: Adenine (A) and Guanine (G). Pyrimidines: Cytosine (C), Thymine (T) (only in DNA), and Uracil (U) (only in RNA)
A Pentose Sugar: Deoxyribose (in DNA) - lacks an oxygen atom at the 2’ position. Ribose (in RNA) - contains a hydroxyl group at the 2’ position.
One or More Phosphate Groups: Phosphate groups are attached to the 5’ carbon of the sugar. The presence of one, two, or three phosphate groups (mono-, di-, or triphosphates) influences the nucleotide’s function (e.g., ATP, GTP).
Biologic synthesis of nucleotides#
De novo nucleotide synthesis:#
Definition: This is the process by which cells synthesize nucleotides from basic molecular precursors such as amino acids, carbon dioxide, and ribose-5-phosphate.
It involves complex, multi-step enzymatic pathways.
Purines (adenine and guanine) and pyrimidines (cytosine, thymine, and uracil) are synthesized via distinct pathways.
In purine synthesis, the nucleotide is built onto ribose-5-phosphate in a stepwise manner, while pyrimidine synthesis involves forming a pyrimidine ring first and then attaching it to ribose-5-phosphate.
This pathway is energy-intensive and is used primarily when there are insufficient nucleotide levels in the cell.
Salvage nucleotide synthesis:#
Definition: This is the process by which cells recycle pre-existing nucleotides, nucleosides, and nitrogenous bases that are released during the breakdown of DNA and RNA.
It is an energy-efficient process compared to de novo synthesis.
Salvage pathways convert free purines and pyrimidines back into nucleotides by attaching them to ribose-phosphate.
Examples include the reuse of hypoxanthine and guanine in purine salvage, facilitated by enzymes like HGPRT (hypoxanthine-guanine phosphoribosyltransferase).
This pathway is critical in tissues with low capacity for de novo synthesis, such as the brain.
Direct nucleosidation abiotic nucleotide synthesis#
Retrosynthetic analysis of RNA involves breaking down the RNA molecule into its constituent nucleotides. Each nucleotide can further be broken down into its components: phosphate and nucleoside. The nucleoside can then be broken down into (D)-ribose and a nucleobase (A, C, G, T, or U). Thus, sugar and nucleobase are the two components that must be synthesized separately and then assembled together to form a nucleoside.
IMPORTANT: Retrosynthetic analysis vs RNA degradation:
RNA degradation is a process that naturally occurs in cells or in the natural environment. It involves the breakdown of RNA molecules, often producing products such as 2’3’-cyclic phosphates at the ends of RNA fragments.
Retrosynthetic analysis is a chemical methodology used to design synthetic pathways for RNA. In this process, RNA is conceptualized as being constructed starting from a 5’-phosphate precursor.
Abiotic sugar synthesis#
Butlerov’s sugar synthesis refers to a method developed by the Russian chemist Aleksandr Butlerov in the mid-19th century, known as formose (formose is a portmanteau of formaldehyde and aldose) reaction. This reaction describes the synthesis of sugars (mainly aldoses) from formaldehyde. Breslow discovered the autocatalytic cycle in Butlerov’s sugar synthesis in the mid-20th century. The C2 sugar, glycolaldehyde, acts as an autocatalyst in the synthesis of longer sugars (Fig. 5.6).
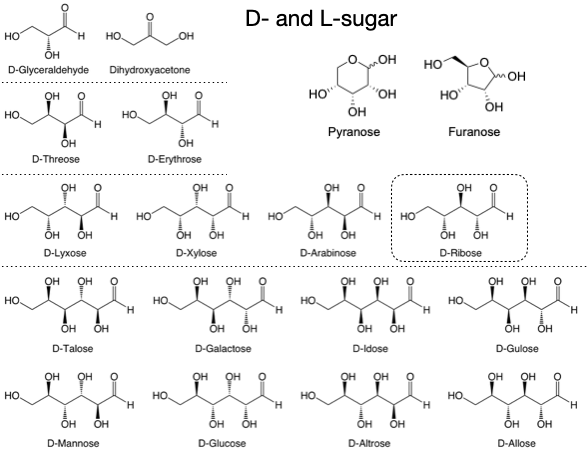
Fig. 5.6 All possible sugars synthesis from Formose reaction, only the structures of D-sugars shown here.#
Disadvantages of the formose reaction:
The products are a mixture of all C2-C6 sugars, with only about 1% of ribose.
The reaction time is a critical factor in controlling the yield, structure, and distribution of products, due to the complexity of the reaction kinetics.
The reaction requires a high concentration of formaldehyde, which would be almost impossible to achieve under early Earth environmental conditions.
Ribose is unstable in aqueous solutions, making its preservation challenging.
All sugars in the product mixture exist as both D- and L-forms.
Improvement of formose reaction: Several inorganic ions have been found to enhance the yields of pentoses (mainly ribose) in the formose reaction, including lead, borate, and silicate.
