5.8. Microbial ecosystems and their geobiological expression#
Professor: Nick Butterfield (Department of Earth Sciences)
Learning objectives:
Understand microbes, microbialites and the evolving Earth system
Understand microbial diversity, distribution and biogeochemical signatures
Be familiar with the geobiological expression of a (non-uniformitartian) microbial world
Introduction#
Life appeared surprisingly early on Earth, but how did it end up in its current form? Biological evolution to be sure, but neither biology or evolution operate in a vacuum. The planet itself was a central part of the action, offering ‘habitability’ in the first instance, but also a set of circumstances that could constructively ‘co-evolve’ to yield both novel lifeforms and novel, biologically modulated environments. On the planetary side this involved the early establishment of plate tectonics and its long term recycling of rocks, volatiles and nutrients. Life rapidly capitalized on the situation, generating a fantastic diversity of metabolic pathways with which to maximize its reach, initiating the planet’s biogeochemical cycles. Over time, the accompanying geobiological feedbacks introduced further novelty, and further opportunities for co-evolutionary expansion.
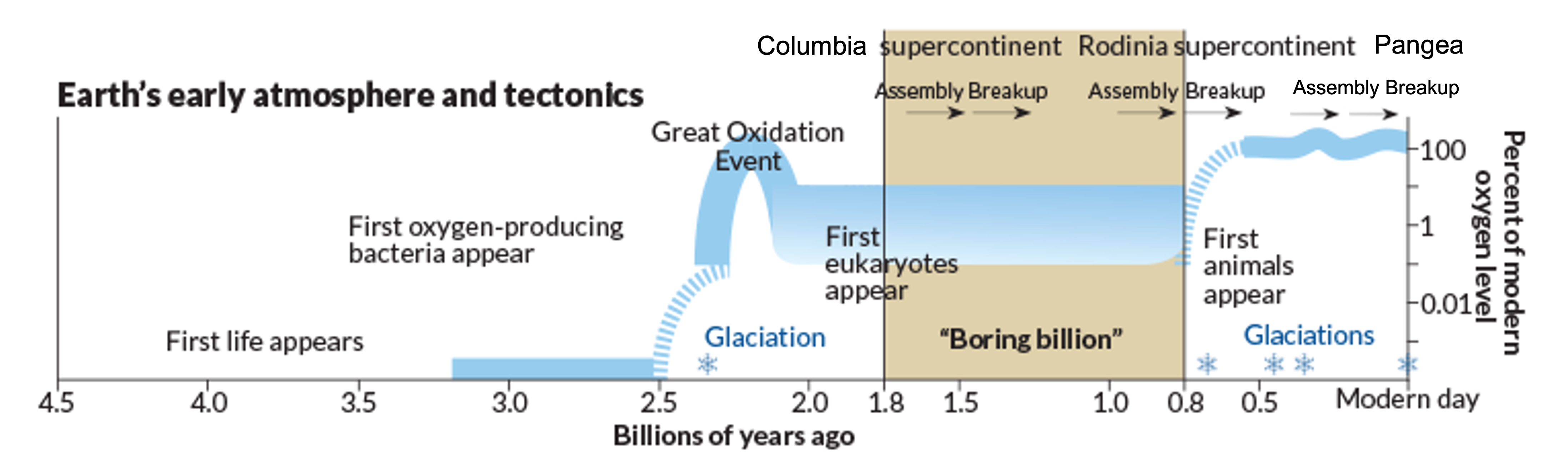
Fig. 5.39 Schematic account of the co-evolving lithosphere, biosphere, and atmosphere over the course of Earth history.#
Photosynthesis, respiration and the Great Oxidation Event#
Carbon-based life requires both a source of chemical energy and a source of reduced carbon. The energy is generated through the transfer of electrons during oxidation-reduction (redox) reactions, and captured in the high-energy bonds of ATP (adenosine triphosphate). The reduced carbon component is supplied either autotrophically, via the ‘fixation’ of environmental CO2, or eterotrophically by consuming someone else who has done it for you. Because CO2 is relatively electropositive (energy-poor) and oxidized, autotrophy requires the input of both energy and electrons.
The vast majority of fixed carbon in the modern biosphere derives from photosynthesis, based on chlorophyll or bacteriochlorophyll. Photosynthesis involves two more or less independent processes:
the ‘light reactions’ which involve the extraction of electrons from external electron donors, powered by solar radiation; and,
the ‘dark reactions’ where ambient O2 is reduced to yield sugars using the chemical energy and reducing power generated by the light reactions.
The removal of electrons from external donors necessarily yields an oxidized ‘waste product’ – which can be recycled as an electron acceptors in the reverse respiration reactions (see below). In the case of oxygenic photosynthesis water is the electron donor and the waste product is molecular oxygen – an extraordinary process given the thermodynamic challenge of oxidizing water at room temperature… By contrast, anoxygenic photosynthesis harvests its electrons from much more amenable, electronegative sources such as hydrogen sulfide (H2S) or ferrous iron (Fe2+). There is a trade-off, however: whereas water is ubiquitous at the Earth’s surface, other electron sources are inevitably localized and limiting (Ward et al. 2019). At least in the long run, oxygenic photosynthesis was the only way for biology to take control of the planetary thermostat.
Oxygenic photosynthesis is the only significant source of molecular oxygen on Earth, but can only accumulate through the long-term sequestration/burial of the reduced organic component (preventing the reverse reaction to CO2 and H2O). At the same time, atmospheric buildup will be delayed by the oxidation of other reduced phases in the surface system, including (tectonically delivered) hydrothermal and volcanic inputs. As such, the appearance of an oxygen-rich atmosphere will have depended not only on the biological evolution of oxygenic photosynthesis, but also the (physical) evolution of the oceans and solid Earth, not least the establishment of large stable cratons with a capacity to accumulate and store sedimentary organic carbon on a planetary scale.
Unlike it anoxygenic counterparts, oxygenic photosynthesis has evolved only once on Earth – in Cyanobacteria. Even so, there is ongoing debate over its evolutionary first appearance. What we do know for sure is that oxygen levels rose from less than \(10^{-6}\) of present-day atmospheric levels (PAL) to more than \(10^{-2}\) PAL during the early Palaeoproterozoic (~2.4 to 2.2 Ga) ‘Great Oxidation Event’. The GOE can be recognized on the basis of contemporaneous palaeosols (fossil soils), presence/absence of detrital pyrite and uraninite, numerous geochemical redox proxies and, most convincingly, the mass-independent fractionation of sulfur (MIF-S). It is also associated with the first major glaciations on Earth (potentially the collapse of an early methane-based greenhouse), the first appearance of continental red beds, and a major (albeit problematic) perturbation in \(\delta^{13}\)C the Lomagundi positive isotope excursion.
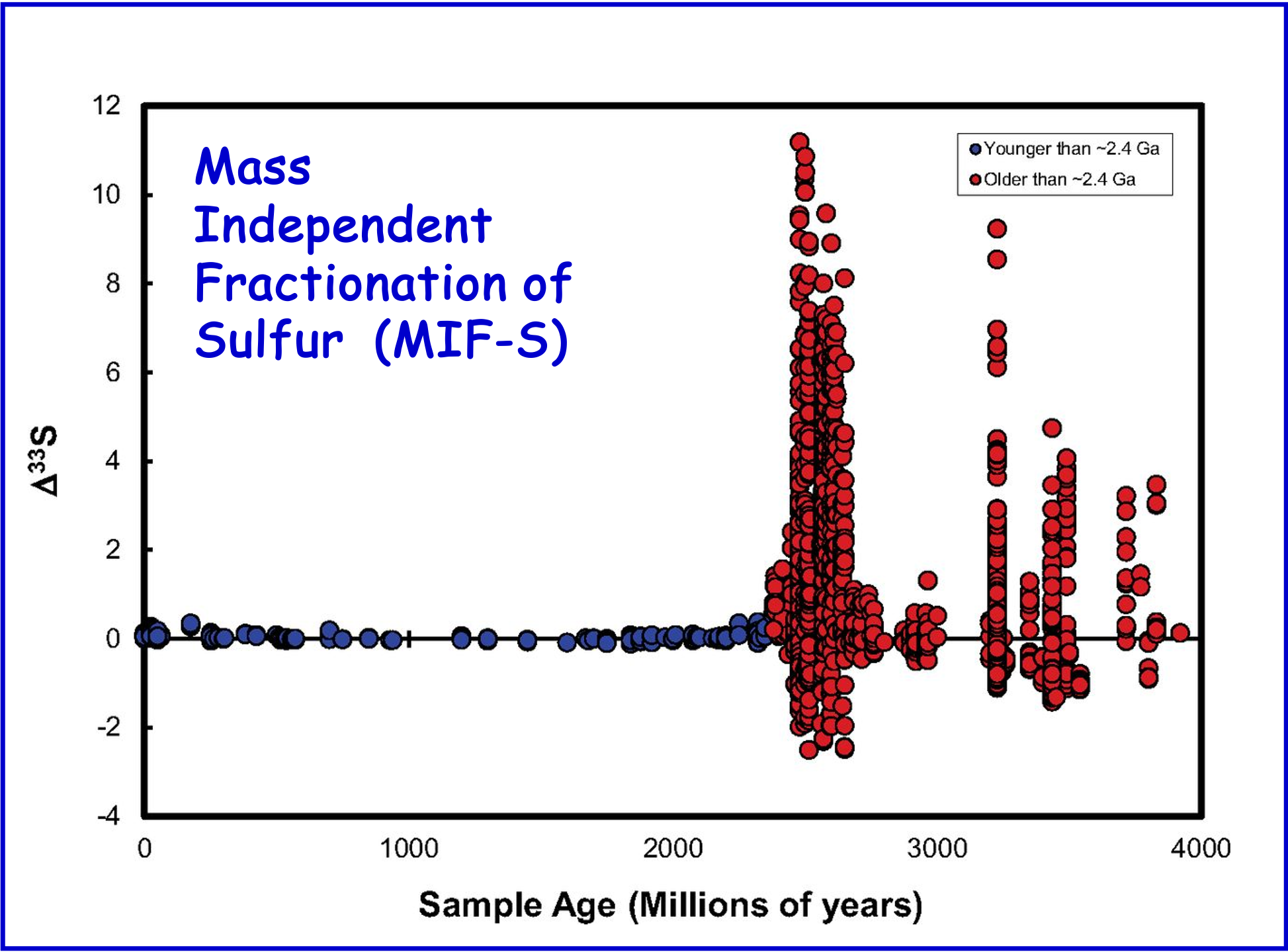
Fig. 5.40 The mass independent fractionation of sulfur isotopes over geological time.#
One of the most conspicuous features of late Archean and early Proterozoic sedimentary systems is the development of Banded Iron Formation (BIF) – represented by enormously expansive accumulations of finely laminated oxidized iron. The traditional model for explaining BIF invokes high levels of reduced hydrothermal iron (Fe2+) in a stratified ocean coming into contact with oxygenated surface water – and precipitating out as insoluble (Fe3+). There is, however, an intriguing alternative that does not require the presence of molecular oxygen – with obvious implications for interpreting planetary oxygenation: rather than being an indirect effect of oxygenic photosynthesis, BIF could simply be the by-product of iron-based anoxygenic photosynthesis – photoferrotrophy.
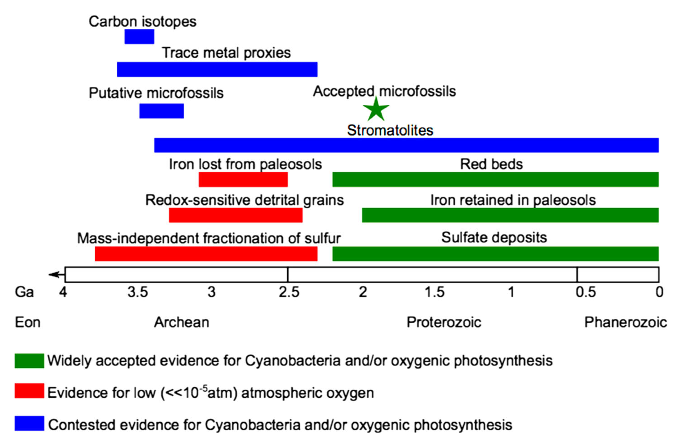
Fig. 5.41 A summary of the lines of evidence documenting the early Proterozoic rise of an oxygen-rich atmosphere - the ‘Great Oxidation Event’.#
Whatever the particular mode(s) of early autotrophic C-fixation, the resulting sugars provided the energy to drive cellular metabolism. Once again (but in reverse), these are redox reactions where the amount of chemical energy/ATP generated is directly related to redox potential of the terminal electron acceptor (TEA). Oxygen, for example, is an exceptionally strong electron acceptor capable of producing 34 moles of ATP from 1 mole of glucose (= aerobic respiration) – vs. less than half of that where Fe(OH)3 is the TEA, and half again in the case of SO4-2 (sulfate reduction). In the absence of any TEA, just two moles of ATP are produced via fermentation/glycolysis.
Early evolution of Eukaryotes#
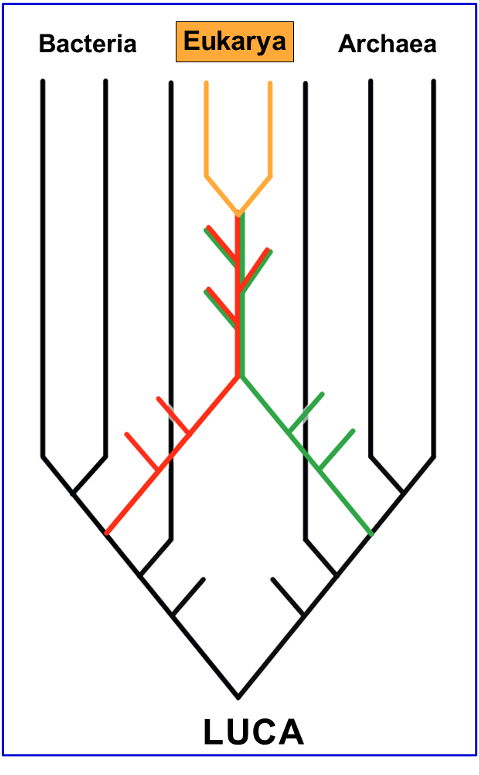
Fig. 5.42 Schematic tree of life charting the emergence of Eukarya.#
The evolution of oxygenic photosynthesis opened the door to aerobic respiration and fundamentally new evolutionary opportunities, not least the rise and radiation of eukaryotes. This ‘third domain’ emerged via a symbiotic association of two ancestral cells – one archaeal and one bacterial – leading to the Last Eukaryotic Common Ancestor (LECA) and, eventually, to the Plants, Animals, Fungi and protists that dominate and define the modern biosphere.
Recognizing eukaryotes in the early fossil record is a challenge. There is no shortage of microfossils in the early Proterozoic, but most are morphologically simple and taxonomically unresolvable. Large cell-size has commonly been used as measure of eukaryotic affiliation but fails on various fronts: not only are there extant examples of giant extant prokaryotes (e.g. mm-sized Thiomargarita), but also any number of colonial aggregations that could be confused as single cells.
The most promising place to search for early eukaryotes is in a waste-basket category known as acritarchs – organic-walled microfossils of unknown taxonomic affinity. Although little can be resolved from simple sphaeromorphic forms, the morphological elaboration exhibited by acanthomorphic acritarchs can be directly related to the presence of a dynamic cytoskeleton – an exclusively eukaryotic attribute. The oldest well-documented records of acanthomorphic acritarchs are Tappania and Shuiyousphaeridium from the late Palaeoproterozoic (~1630 Ma) of India and North China. There is no reason to assume these relate to the evolutionary first appearance of eukaryotes, but they do set a reliable minimum age for the group.
The early Proterozoic record also includes a range of macroscopic/multicellular problematica, that may or may not be eukaryotes. One of the earliest is Grypania, a conspicuously coiled, sometimes septate, macroscopic filament reported from 1900–1400 Ma mudstones. Although originally interpreted as multicellular green algae, there is nothing diagnostically algal, or even eukaryotic about these fossils. Indeed, it’s been suggested they might be nothing more than ‘giant’ cyanobacteria.
References#
Butterfield NJ. 2015. Proterozoic photosynthesis – a critical review. Palaeontology 58, 953–972.
Butterfield NJ. 2015. Early evolution of the Eukaryota. Palaeontology 58, 5–17.
*Catling DC, Zahnle KJ. 2020. The Archean atmosphere. Science Advances 6, eaax1420.
Crockford PW, et al. 2023. The geologic history of primary productivity. Current Biology 33, 1–10.
