4.5. Astrochemistry: From the Insterstellar Medium to Comets#
Lecturer: Paul B Rimmer (Department of Physics, Cavendish Laboratory)
In the next two lectures, we will cover astrochemistry proper, and how astrochemical knowledge and methods may help bridge our knowledge of chemistry in space to the chemistry of life.
Key Concepts for L9 and L10: - Chemical reactions in the Interestellar Medium - The First Chemical Species in our Universe - Chemistry of Photon-Dominated Regions (PDR’s) - Disk Chemistry and Comet Chemistry
Lecture overview#
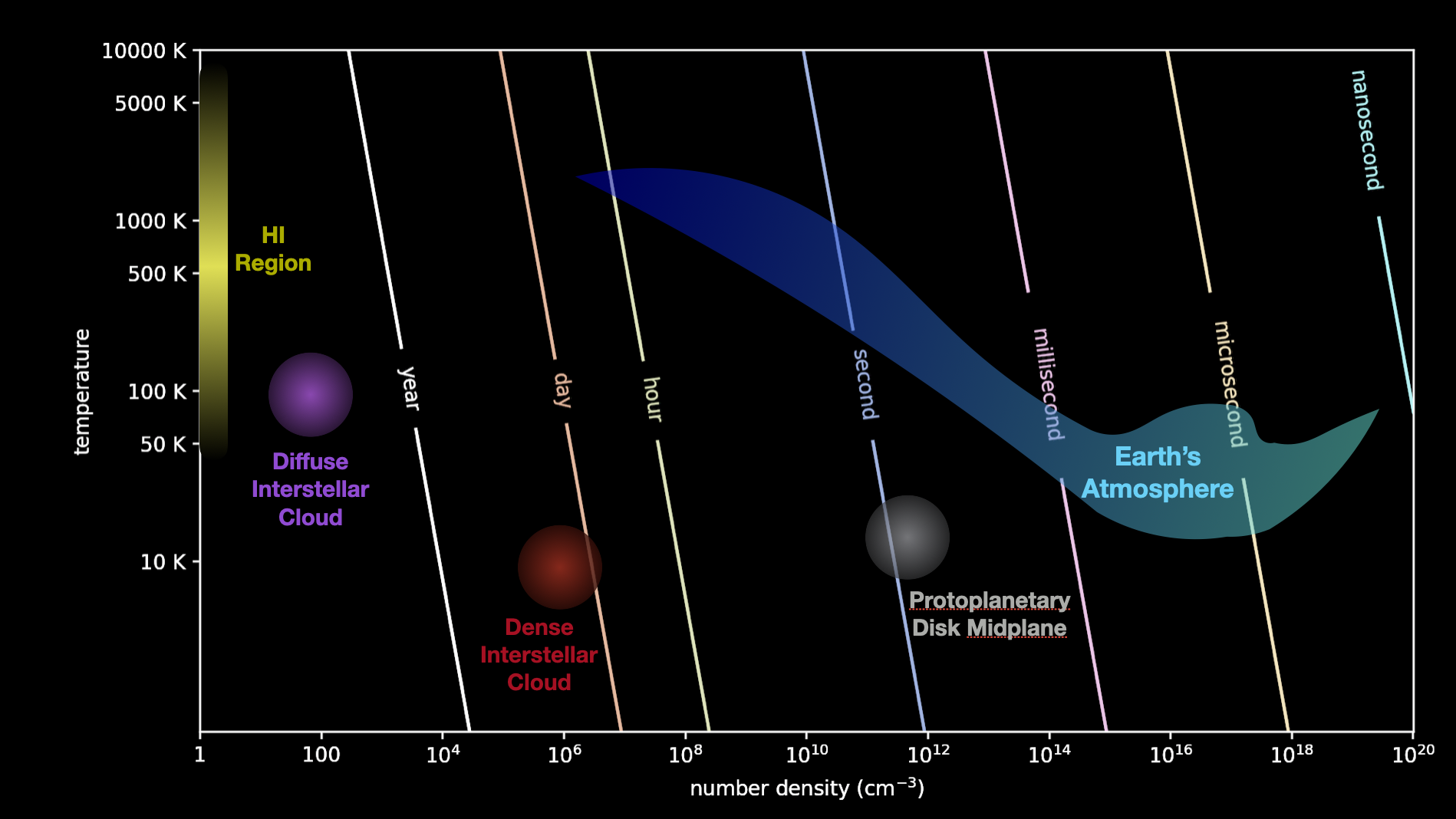
Fig. 4.43 Timescales for Molecular Collisions: An illustration of how long a given atom would have to wait (order of magnitude) before colliding with another atom, as a function of temperature and number density of the environment. Highlighted are certain astrophysically relevant environments, such as H1 regions, diffuse interstellar medium, dense interstellar medium, protoplanetary disk midplane, and Earth’s atmosphere.#
In this lecture, we will go from the interstellar medium to comets, looking at the mechanisms and the overall story astrochemistry tells us about our chemical origins, and the rich chemistry that permeates our galaxy and universe.
In the next lecture, we will connect these methods and this story to the far-less-well-developed story of how life may have originated on Earth, and how its origins may show us something about the possibility of life on other planets (and vice versa).
Chemistry in the Interestellar Medium#
Astrochemistry is the study of chemistry in space. Most of the chemistry we will look at takes place in the interstellar medium, the space between stars, and is divided into chemistry in:
The diffuse interstellar medium
The dense interstellar medium
The disk of gas and dust surrounding a proto-star or star
As you can see from the above figure, even this list involves a range of temperatures spanning around an order of magnitude, and a range of number densities spanning ten orders of magnitude!
The “chemistry” part of astrochemistry deals with the elements present in space, the bonds those elements form, how those bonds are formed, and how quickly they are formed (i.e. the kinetics).
The elements we are working with are largely those elements that arose from the big bang, along with a smattering of heavier elements (astronomers tend to call these ‘metals’) from stellar explosions and other high-energy events.
The “astronomer’s periodic table” is a much sparcer landscape than the chemist’s periodic table, and can be seen in the figure below.
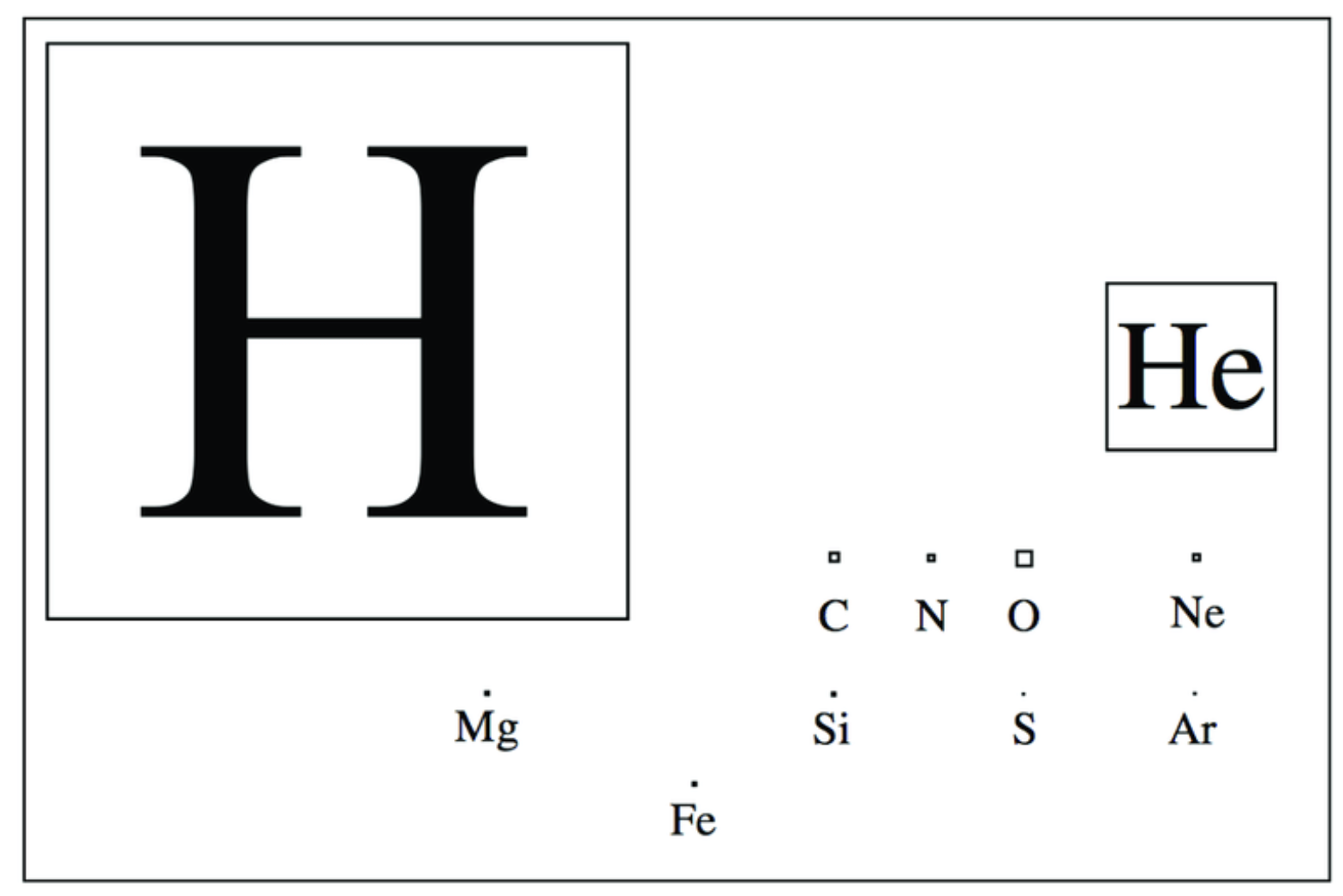
Fig. 4.44 The Astronomer’s Periodic Table: Showing the most abundant elements, with the squares loosely representing their relative abundances. Courtesy of Benjamin McCall (2001).#
These elements and their abundances indicate that the chemistry of the interstellar medium will be hydrogen-rich, and that most oxygen, carbon, and nitrogen atoms will be hydrogen-saturated. This turns out not to be the case because of chemical kinetics: the nature of reactions that tend to take place in the interstellar medium.
As you can see from the below figure, there are many exotic molecules, long aliphatic carbon chains, all the way up to C\(_{60}\), that have been observed in the interstellar medium, most of them in the dense high-energy environment near the black hole at the center of our galaxy: Sgr B2.
These chemical species admit a great deal of chemical complexity, and some resemble organic precursors to the molecules used by life. These chemical species arose from sequences of dozens to hundreds of chemical reactions. What determines the chemical environment of space?
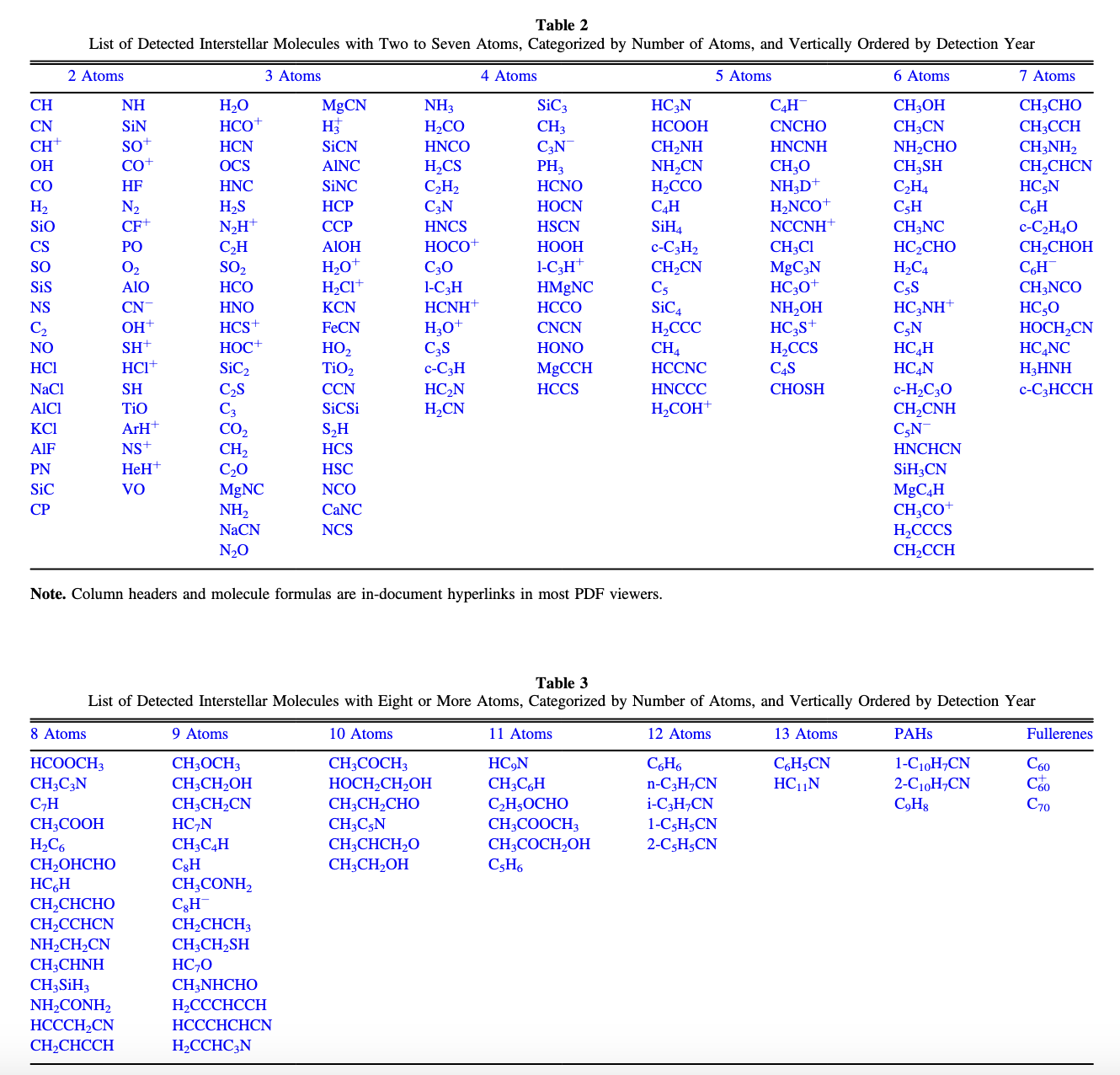
Fig. 4.45 List of Chemical Species found in the ISM: Up-to-date as of 2022, from McGuire (2022).#
First, for a reaction to take place, two atoms need to find each other. For the diffuse or dense interstellar medium, that means waiting days or even years for a single collision, depending on the actual density and the relative concentrations of the different atoms.
Even if atoms hit each other, to form radicals, and eventually stable molecules, not every collision will result in the formation of a stable chemical bond. Different reactions will require energy to proceed from reactant to product, and at 10 or even 100 K, there’s not that much thermal energy to be had. In these environments, chemical reactions must either be driven by far-from-equilibrium processes, such as interaction with ultraviolet or X-ray light, gamma rays, or cosmic rays; or they must be exergonic and barrierless, what you may have heard called spontaneous reactions.
Consider the energetic landscapes for three different candidate reactions for A + B \(\rightarrow\) X + Y, in the figure below.
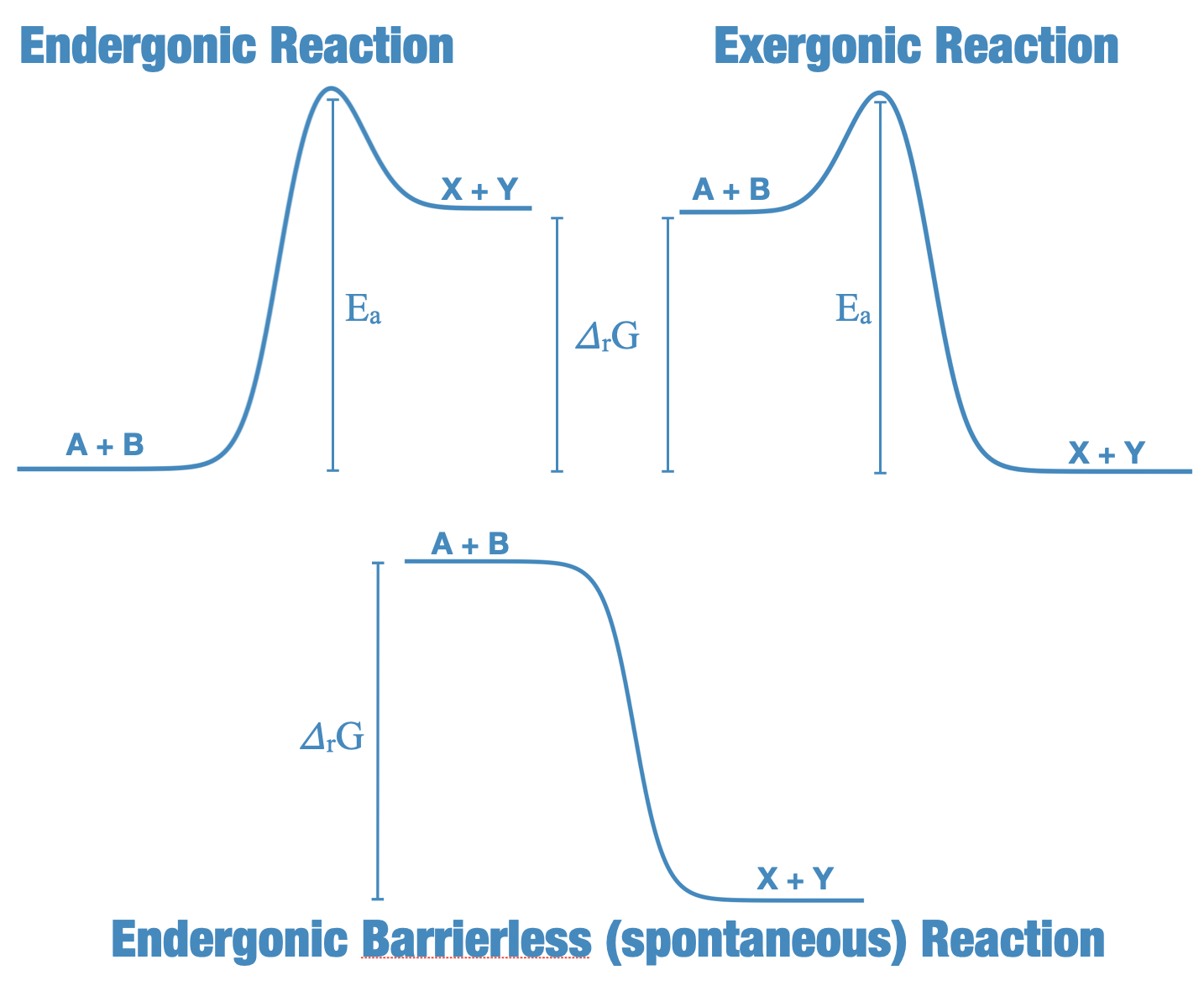
Fig. 4.46 Reaction Energetics: We see cartoon examples of two reactions with barriers, an endergonic and exergonic reaction, and at the bottom, a barrierless exergonic reaction. The change in Gibbs free energy over the reaction, \(\Delta_r G\) (eV) is shown, as is the activation energy (\(E_a\), eV).#
Besides the reactions involving energetic particles, like photons and cosmic rays, the two most common reacations in the insterstellar medium are ion-neutral reactions and dust surface reactions. Let’s take a brief look at each in turn.
Ion-neutral reactions involve an ion crashing into a neutral molecule, and tend to involve the formation of a new ion and neutral moleule, such as:
There are many different names for these reactions, depending on what’s happening, whether charge exchange of the re-arrangement of some of the bonds in A and B from the AB\(^+\) complex to form X\(^+\) and \(Y\). We can still estimate the rate constant, or rate coefficient, of this and other reactions.
For this, we take that the rate constant is equal to:
where \(\sigma\) (cm\(^2\)) is the collisional cross-section, \(v\) (cm/s) is the velocity at the collision, and \(\alpha\) is hte probability that the two molecular species, colliding at that velocity, react to form a given set of reactants. The temperature dependence for the rate constant comes in sometimes for \(\sigma\), becuase the collisional cross-section can change with temperature. The average velocity also changes with temperature, and even in the interstellar medium, the assumption of Maxwell-Boltzmann statistics is typically valid (why do you think this is?), so the average velocity is often just representated as:
and is separated out of the average (why is this a fair thing to do? How would you get around doing this?).
In the case of an ion-neutral collision with an ion and a polarizable molecule, we can see that the polarized molecule experiences an attractive force as a function of intermolecular distance, \(r\) (m), of \(F = P \cdot \big(dE/dr\big)\), where \(P = \alpha_N E\) (C m) is the dipole moment, and \(\alpha_N\) (C m\(^2\)/V) is the polarizability, and \(E\) (V/m) is the electric field. Given a Coulomb field, \(E = kq/r^2\) where the charge here is typically equal to the charge of a single electron, \(q = 1.6 \times 10^{-19}\) C, and \(k = 8.99 \times 10^9\) N m\(^2\) C\(^{-2}\); we can then solve for the force:
Given that \(F = -dU/dr\), we can represent the potential energy of the polarized molecular species in the Coulomb field as:
We approximate \(\sigma = \pi r_0^2\), where \(r_0\) (m) is the effective collisional radius, namely, the value \(r\) takes when the potential energy is equal to the kinetic energy:
Try this out for yourselves. This will give:
Because we have that \(k = \langle \sigma v \rangle\), and \(\sigma\) depends on \(1/v\), ion-neutral reactions are independent of velocity, and therefore temperature. Moreover, if we put numbers into each of these variables, and use typical values for the polarizability, we find that, for ion-neutral reactions:
Compare this to the rate constants from the Atmospheric Chemistry lectures: would reactions with this magnitude rate constant be fast or slow?
Because ion-neutral reactions are temperature-independent, they can proceed even at very low temperatures, such as the temperatures in dense interstellar clouds, e.g. 10 K. Ion-neutral reactions, if they happen at all, are typically spontaneous. No barriers to worry about.
A particular kind of neutral reaction, even when it would be spontaneous, has a terrible time in the interstellar medium. This is a termolecular reaction, when two atoms or molecules come together to form a single complex.
The reason this is a difficult reaction is because the single complex never has exactly the same energy as the two atoms or molecules going in. If it has more energy, then this acts as a barrier, and these barriers are very hard to overcome at such low temperatures. But even if the reaction is exergonic, what will happen is illustrated below:
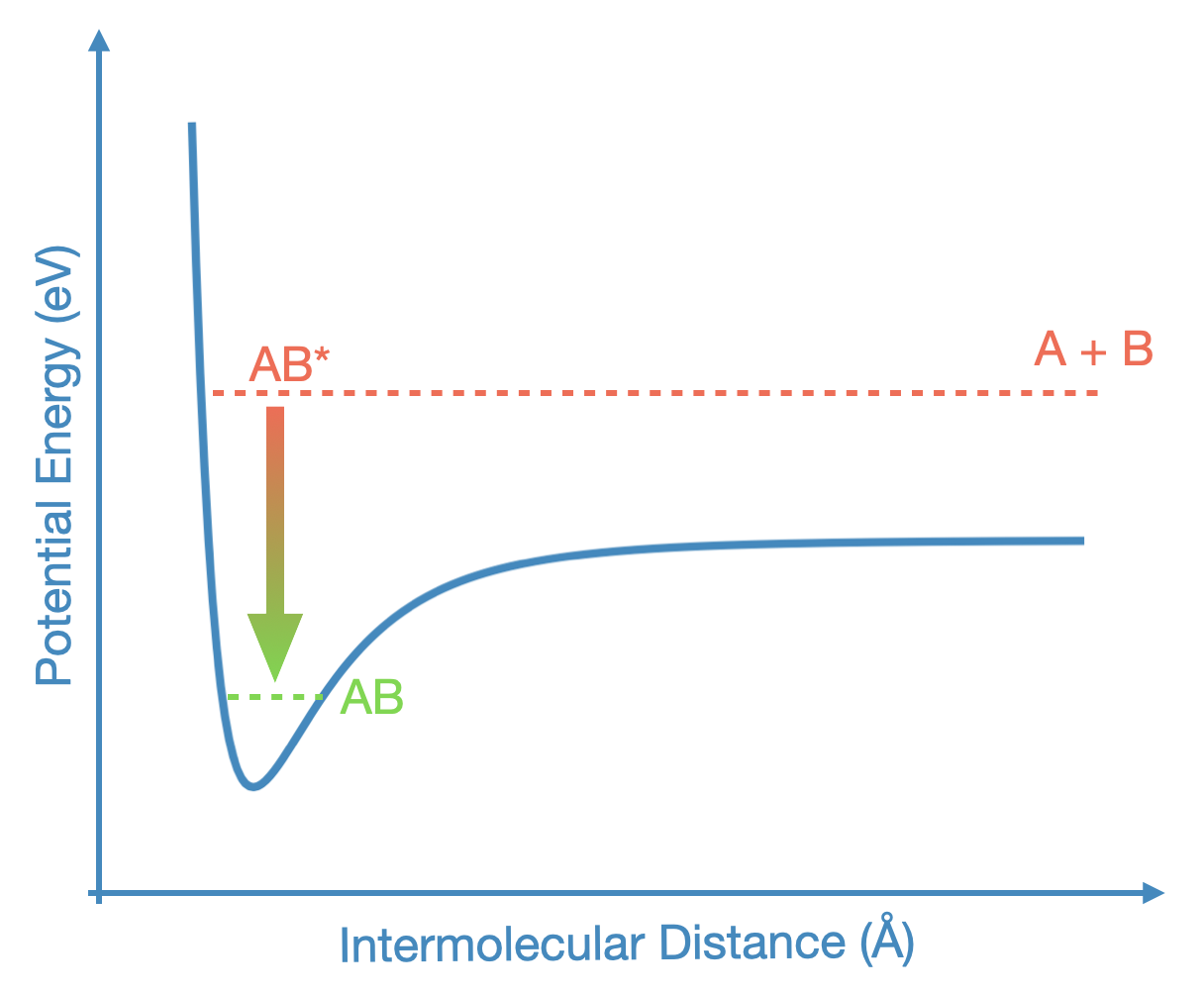
Fig. 4.47 An Energetic Challenge for Termolecular Reactions: This shows two chemical species, A and B, entering into a potential well. This particular potential energy curve, as a function of the intermolecular distance, is a Lennard-Jones potential, of the form \(1/r^{12} - 1/r^6\). When above the well, teh atoms form a complex, AB*, but this complex will quickly just break apart into A + B as the species leave the well, unless they can relax into a lower energy state. This could happen spontaneously, by release of a photon (\(h\nu\)), or the energy could be released into some third body, like a dust grain.#
A + B come together to form a complex, AB*, and this complex now has too much energy to remain bound. If there were a third body that would collide with AB*, it could take away that excess energy, allowing \({\rm AB^* \rightarrow AB}\). But in the interstellar medium, there is no third body that will find AB* before it simply breaks apart. It is possible for AB* to spontaneously release a photon:
This reaction is known as radiative association. The most abundant molecule in our universe is H\(_2\), and we might think H\(_2\) could have formed by this radiative association. The rate constant for radiative association of \({\rm H + H} \rightarrow {\rm H_2} + h\nu\) is \(< 10^{-26}\) cm\(^3\) s\(^{-1}\). This means that the time it would take to convert a significant amount of H into H\(_2\) in a dense region is:
Around 100x the age of the universe!
The way H\(_2\), and many other molecules in the interestellar medium, likely form, involves dust surface chemistry. There’s a lot of dust in the dense interstellar medium, it is largely what makes molecular clouds look so dark. At cold temperatures, chemical species, atoms and molecules, like atomic hydrogen, can attach themselves onto the surface of the grain, like atomic-hydrogen-ice, but they will wander on that surface, weakly bound as they are, until they run into another hydrogen atom. Then they can join together, and release the extra energy into the grain. Like the picture below:

Fig. 4.48 Interstellar Dust Surface Chemistry: A snap-shot of dust surface chemistry in the interstellar medium, forming water in this case (along with other molecules and radicals). Taken from van Dishoeck & Lamberts NOVA Video (Leiden University).#
For a nice review of both gas-phase and grain-surface chemistry, see Herbst (2001).
Once H\(_2\) is formed, a path is now opened to more complex chemistry, including organic species. One path to these organic species involves cosmic rays. Cosmic rays can ionize H\(_2\), opening up ion-neutral chemistry, which we already argue is more effective under interstellar conditions.
Let’s look at a simple sequence of reactions:
Other reactions with H\(_3^+\) and atomic carbon can lead to rich interstellar chemistry, see Larsson et al. (2012). This chemistry depends on the concentration of H\(_3^+\), and the steady-state concentration can be calculated from the rate equations based on the simple series of reactions above.
A great database to search astrochemical reactions is the Kida Database. Another good database is the UMIST Database.
Astrochemistry through the History of the Universe#
The very first molecular species in our universe formed before there were dust grains, because dust grains themselves are molecular, and are made of heavy elements, and would not have formed until the death of other stars. Early in the life of the universe, temperatures were high enough that a good fraction of the hydrogen and helium was ionized. Too early, and any kind of chemistry would have been prohibitive at such high temperatures, but as the universe expanded and cooled, ions could react with neutral atoms, namely:
HeH\(^+\) (or possibly He\(_2^+\)) was the first chemical species in our universe. The radiative association reactions shown here with ion-neutral species are still very slow (\(\sim 10^{-20}\) cm\(^3\) s\(^{-1}\), see Roberge & Dalgarno 1982), but not nearly as slow as H + H radiative association, or most neutral atomic radiative association reactions. This is not fast enough to explain how all H\(_2\) in a dense cloud would have been formed, but it is enough to predict that there would have been significant abundances of various molecular species in the early Universe, before dust grains were formed.
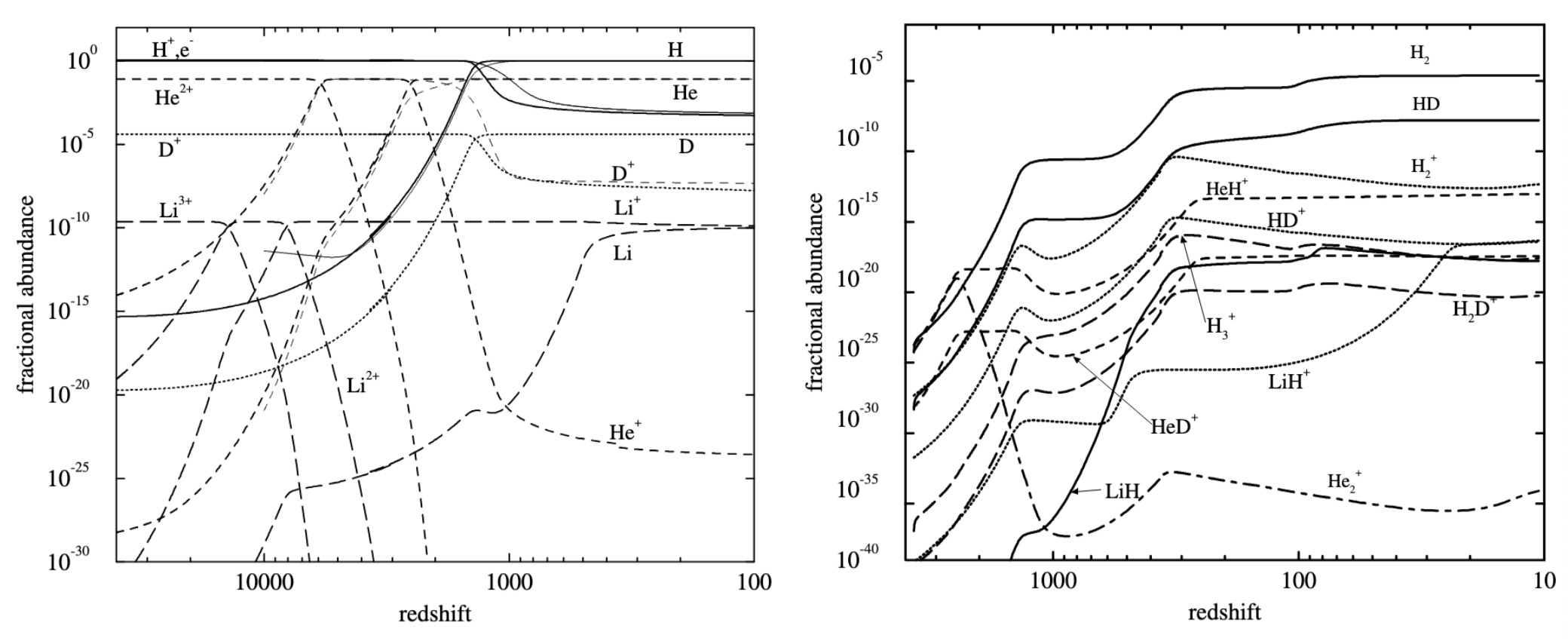
Fig. 4.49 The Molecular History of the Early Universe: Predicted concentrations of atomic and ion abundances, along with the abundances of molecular species, from Lepp et al. (2002).#
Cosmic-ray-driven, X-ray-driven and UV-driven chemistry have been seen in the accreation disks around Active Galactic Nuclei (AGN), with an early tracer being an overabundance of HCN and enhanced HNC/HCN ratios (Harada et al. 2010; Izumi et al. 2016).
This chemistry is also seen in photon-dominated regions (PDR’s), an entire subject of its own, relevant for chemistry in nebulae, such as the Horsehead Nebula and the Orion KL region. See Wolfire et al. (2022).
There is also rich chemistry in protoplanetary disks, throughout their evolution, and there are likely deep links between astrochemistry and planetary formation, given several formation scenarios invoke instabilities that depend on the ionization fraction of the material in the disk. See Öberg et al. (2023), and also this nice illustration below of a disk environment, highlighting different chemically-relevant regions. Below that is an illustration of the striking similarities between disk chemistry and comet ice chemistry.
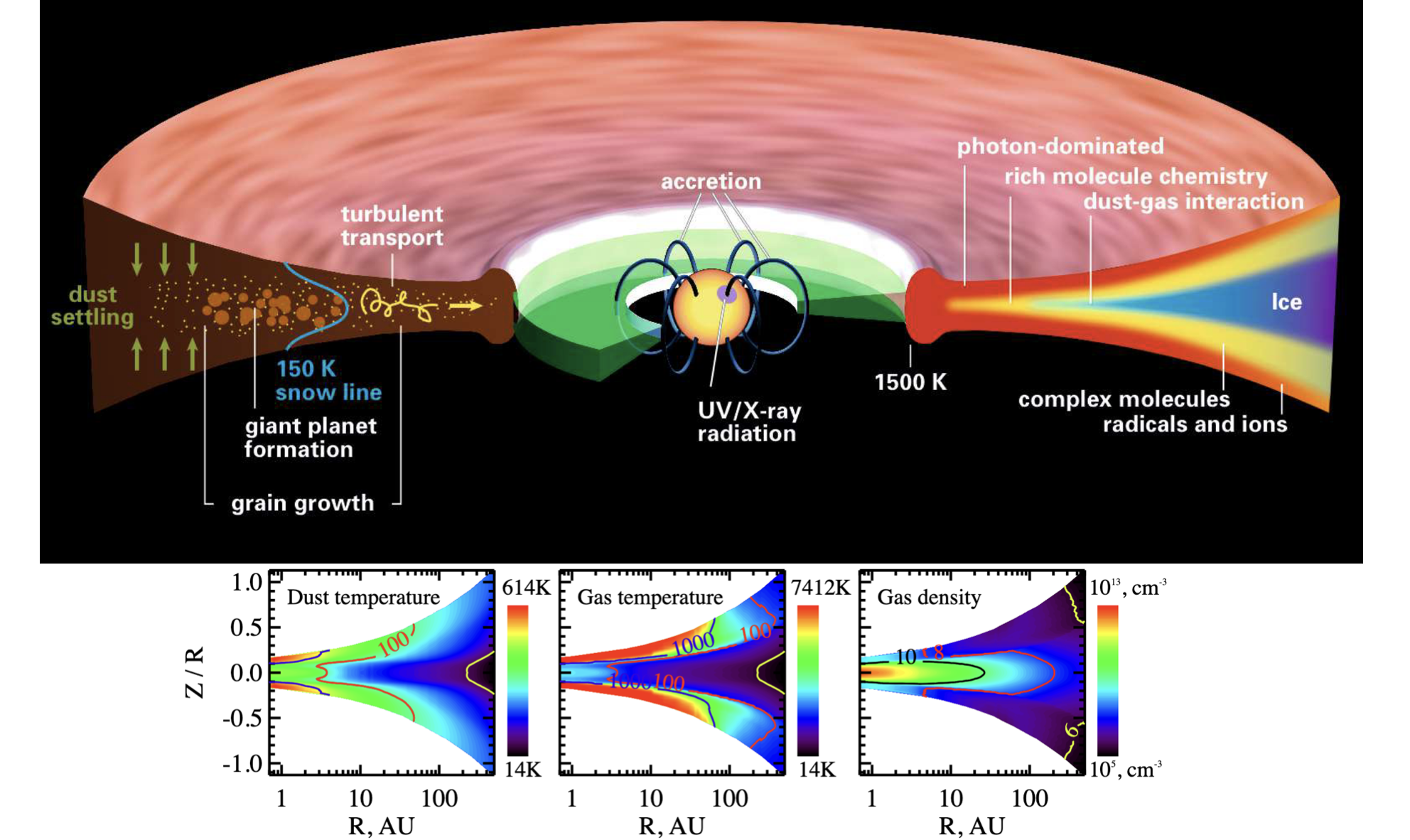
Fig. 4.50 Protoplanetary Disk Chemistry: An illustration of the structure of a protoplanetary disk, highlighting which regions are photon-dominated, which have rich chemistry, which have ice, where snow lines are, and, below, the gas densities, temperatures (gas and dust) of different regions within the disk. From Henning & Semenov (2013).#
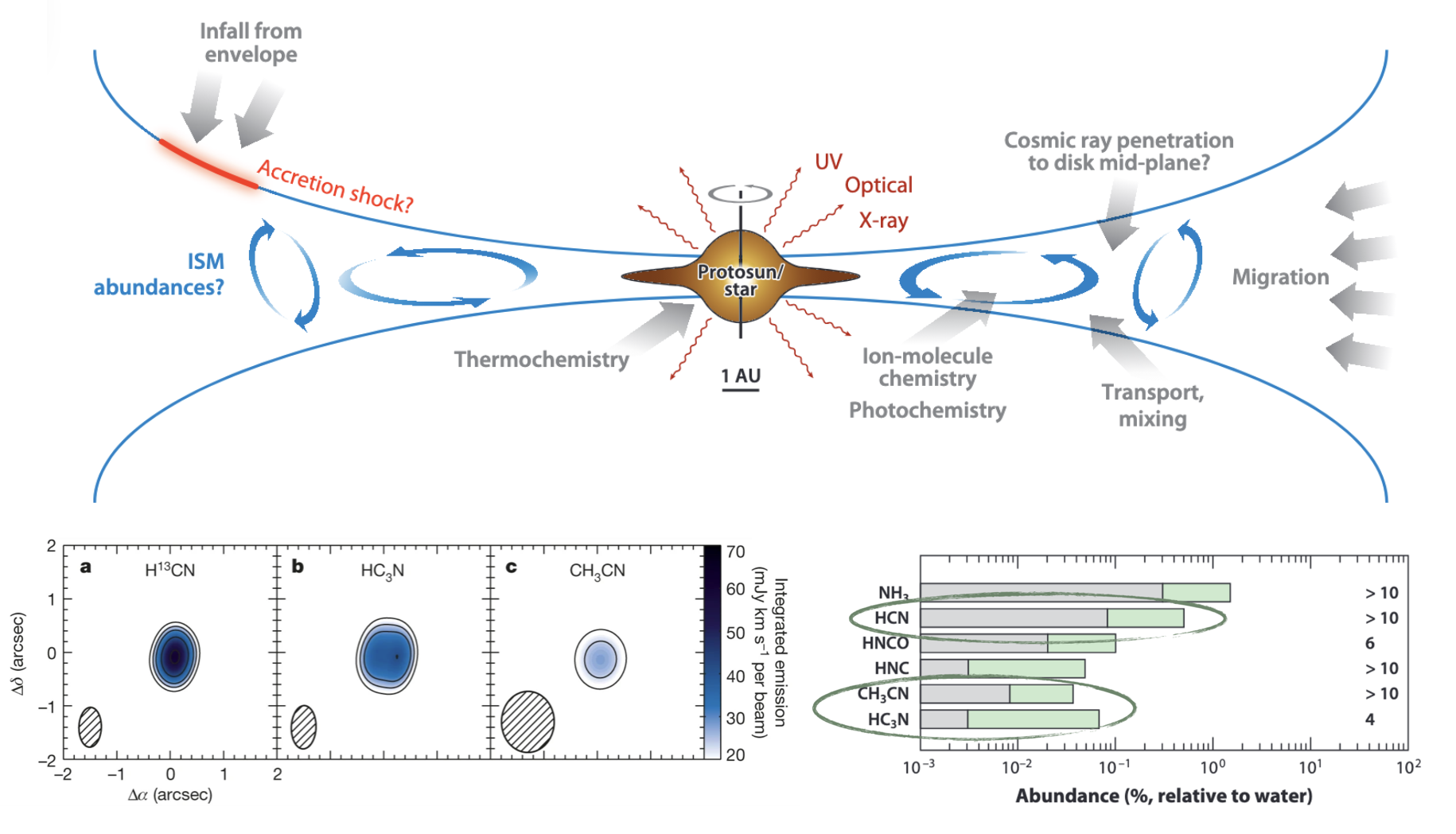
Fig. 4.51 Protoplanetary Disk Chemistry: Comparison of nitrile (CN-triple-bond) chemistry between a protoplanetary disk and comet measurements. From Öberg et al. (2015) and Mumma & Charnley (2011).#
Futher Reading#
Harada, N., Herbst, E. and Wakelam, V., 2010. A new network for higher-temperature gas-phase chemistry. I. A preliminary study of accretion disks in active galactic nuclei. The Astrophysical Journal, 721(2), p.1570.
Henning, T. and Semenov, D., 2013. Chemistry in protoplanetary disks. Chemical Reviews, 113(12), pp.9016-9042.
Herbst, E., 2001. The chemistry of interstellar space. Chemical Society Reviews, 30(3), pp.168-176.
Izumi, T., Kohno, K., Aalto, S., Espada, D., Fathi, K., Harada, N., Hatsukade, B., Hsieh, P.Y., Imanishi, M., Krips, M. and Martín, S., 2016. Submillimeter-hcn diagram for energy diagnostics in the centers of galaxies. The Astrophysical Journal, 818(1), p.42.
Larsson, M., Geppert, W.D. and Nyman, G., 2012. Ion chemistry in space. Reports on Progress in Physics, 75(6), p.066901.
Lepp, S., Stancil, P.C. and Dalgarno, A., 2002. Atomic and molecular processes in the early Universe. J. Phys. B: At. Mol. Opt. Phys, 35, pp.R57-R80.
McGuire, B.A., 2022. 2021 census of interstellar, circumstellar, extragalactic, protoplanetary disk, and exoplanetary molecules. The Astrophysical Journal Supplement Series, 259(2), p.30.
Mumma, M.J. and Charnley, S.B., 2011. The chemical composition of comets—emerging taxonomies and natal heritage. Annual Review of Astronomy and Astrophysics, 49(1), pp.471-524.
Öberg, K.I., Guzmán, V.V., Furuya, K., Qi, C., Aikawa, Y., Andrews, S.M., Loomis, R. and Wilner, D.J., 2015. The comet-like composition of a protoplanetary disk as revealed by complex cyanides. Nature, 520(7546), pp.198-201.
Öberg, K.I., Facchini, S. and Anderson, D.E., 2023. Protoplanetary disk chemistry. Annual Review of Astronomy and Astrophysics, 61(1), pp.287-328.
Roberge, W. and Dalgarno, A., 1982. The formation and destruction of HeH/+/in astrophysical plasmas. Astrophysical Journal, Part 1, vol. 255, Apr. 15, 1982, p. 489-496., 255, pp.489-496.
Wolfire, M.G., Vallini, L. and Chevance, M., 2022. Photodissociation and X-Ray-Dominated Regions. Annual Review of Astronomy and Astrophysics, 60(1), pp.247-318.
