2.6. The origin of the elements#
Professor: Christopher Tout (IoA)
Learning objectives:
Big bang nucleosynthesis.
Energy production in stars, hydrogen burning and catalytic cycles.
Helium burning and alpha elements.
Slow and rapid neutron capture.
The sites of nucleosynthesis.
At the beginning of the Universe matter appeared as protons and neutrons. Early fusion could get as far as helium-4 but not beyond. Everything else requires stars in which heavier elements might be considered as nuclear waste products. Main-sequence stars are fusing hydrogen to helium either directly or by catalytic cycles. Once hydrogen has been exhausted and the temperature has risen sufficiently helium nuclei can fuse to form carbon, oxygen, neon etc., the \(\alpha\) elements. Beyond the iron group fusion no longer releases energy and heavier elements require capture of uncharged neutrons. We examine in which stars and when these processes proceed along with how the enriched material is returned to the interstellar medium whence it can be incorporated in new stars and planets.
Big Bang Nucleosynthesis#
Protons carry a positive electric charge and so repel one another. Neutrons do not and so, when present, can easily react with protons.
\(\rm p + n \rightarrow D +\gamma,\)
where \(\rm p\equiv {^1H}\) represents a proton, n a neutron \(\rm D\equiv {^2H}\) a deuterium nucleus and \(\gamma\) an energetic photon. This is followed by
\(\rm p + D \rightarrow {^3He} + \gamma\)
and
\(\rm D + {^3He} \rightarrow {^4He + p}\)
creating helium-4. Side reactions lead to a small production of lithium, beryllium and boron.
The Universe expanded too fast for all but a tiny quantity of carbon to be created from fusion of helium. Significant carbon and everything else requires stars.
Nuclear Fusion in Stars#
In the 1920s Eddington used \(E = mc^2\) to argue that the fusion of hydrogen to helium could power the Sun over geological timescales. He also argued that the centre of the Sun is not hot enough.
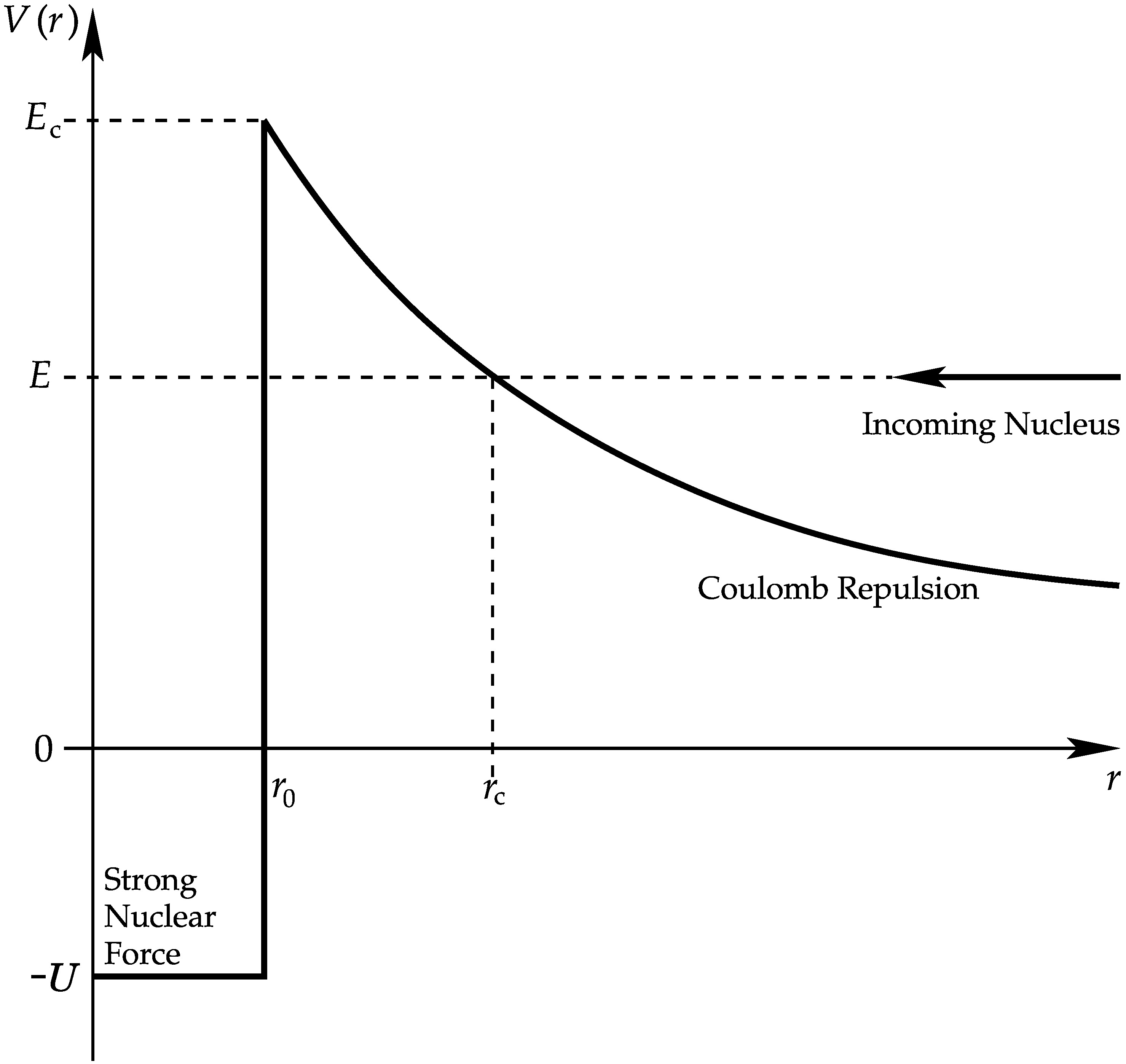
Fig. 2.34 The potential energy \(V\) of two nuclei of relative energy \(E\) separated by \(r\) in their centre of momentum frame.#
At small \(r < r_0\), the effective nuclear radius, the short range strong nuclear force dominates and \(V ≈ −U\). At large \(r\) the Coulomb repulsion dominates with
\(V(r) = {Z_AZ_Be^2\over(4\pi\epsilon_0 r)}.\)
The classical Coulomb barrier could only be overcome if \(E > E_{\rm c}\). The radius at which \(E = V (r > r_0)\) is \(r_{\rm c}\).
Around 1930 Gamov showed it could work with quantum mechanical tunnelling.
The Proton–Proton Chains#
At about \(T = 2\times 10^7\,K\) hydrogen burns by the pp~I chain
\(\rm {^1H} + {^1H} \rightarrow {^2H} + e^+ + \nu,\)
\(\rm e^+ + e^- \rightarrow \gamma,\)
\(\rm {^2H} + {^1H} \rightarrow {^3He} + \gamma,\) \(\rm {^3He} + {^3He} \rightarrow {^4He} + 2{^1H}.\)
The pp~II and pp~III chains occur as side reactions and can generate Li, Be and B in small quantities.
These pp chains dominate in stars with \(M\le 2\,M_\odot\).
For all stars on the main sequence hydrogen burning takes place deep in the core and no change in composition is seen at the surface.
Carbon, Nitrogen, Oxygen Cycle#
At higher temperatures so in more massive stars hydrogen burns by catalytic cycles beginning with the CN and CNO
\(\rm {^1H} + {^{12}C} \rightarrow {^{13}N} + \gamma\)
\(\rm {^{13}N} \rightarrow {^{13}C} + e^+ + \nu_{\rm e}\) \(\rm {^1H} + {^{13}C} \rightarrow {^{14}N} + \gamma\)
\(\rm {^1H} + {^{14}N} \rightarrow {^{15}O} + \gamma\)
\(\rm {^{15}O} \rightarrow {^{15}N} + e^+ + \nu_{\rm e}\)
\(\rm {^1H} + {^{15}N} \rightarrow {^{12}C} + {^4He} + \gamma\)
and sometimes at higher temperatures
\(\rm {^1H} + {^{15}N} \rightarrow {^{16}O} + \gamma\)
\(\rm {^1H} + {^{16}O} \rightarrow {^{17}F} + \gamma\)
\(\rm {^{17}F} \rightarrow {^{17}O} + e^+ + \nu_{\rm e}\)
\(\rm {^1H} + {^{17}O} \rightarrow {^{14}N} + {^4He}\)
The proton capture by nitrogen-14 is the slowest step and so CNO elements tend to be converted to nitrogen during hydrogen burning.
At higher temperatures other such catalytic cycles such as sodium, neon, magnesium are activated but all require the catalysts in place.
Nuclear Binding Energy#
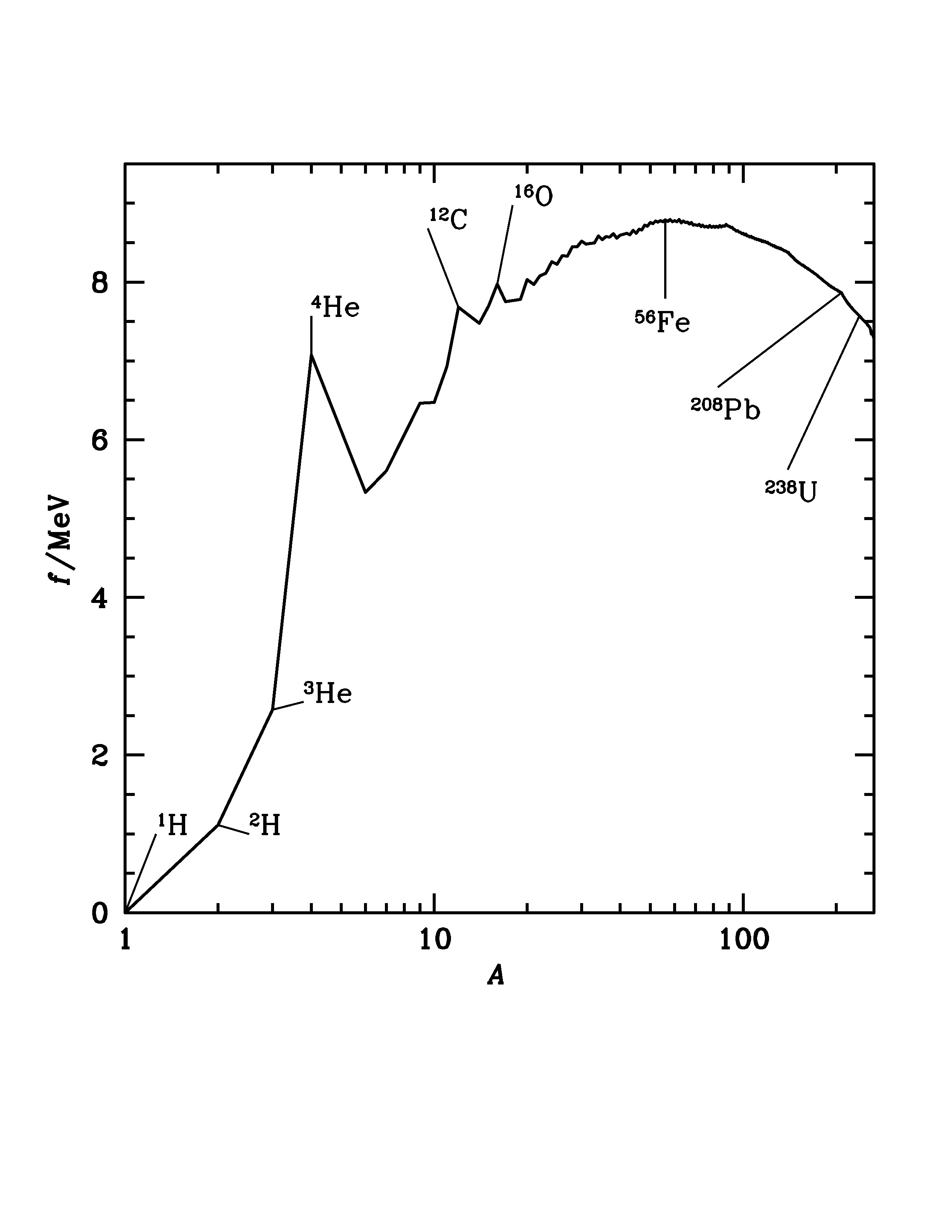
Fig. 2.35 The binding energy per nucleon \(f\) for the most stable isotope of each atomic weight \(A\). Iron-56 occupies the site of the most stable nucleus. Energy can be obtained by fusion of nuclei of lower atomic weight or fission of nuclei of higher atomic weight. Notable peaks occur appear for helium-4, carbon-12 and oxygen-16. Lead-208 is the heaviest non-radioactive nucleus.#
The Triple-\(\alpha\)-Process#
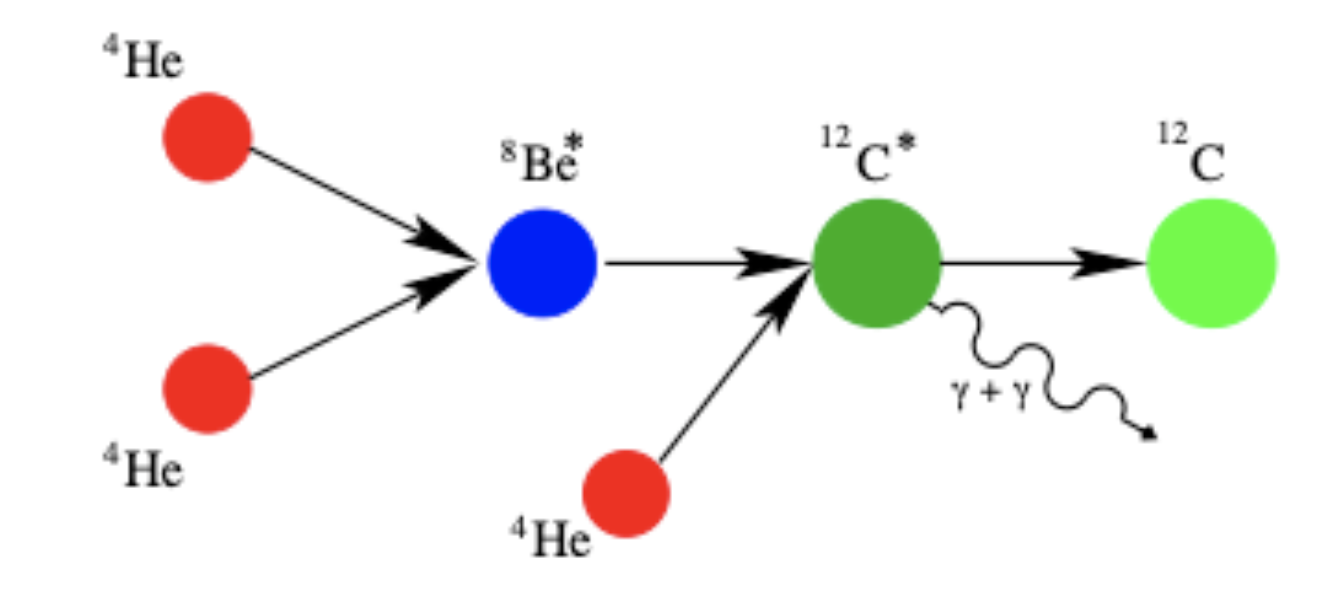
Fig. 2.36 Fusion of helium-4 to carbon-12.#
Otherwise hydrogen burning leaves behind almost pure helium-4 cores in stars. Once the temperature has risen sufficiently (\(T\approx 10^8\,\)K) this \(^4\)He can provide the next source of energy. It happens that there is no stable nuclear configuration with \(Z=4\) and \(A=8\). The reaction
\(\rm {^4He} + {^{4}He} \buildrel\leftarrow\over\rightarrow {^8Be}^{*},\)
is endothermic and so easily reversed. The unstable nucleus \(\rm ^8Be^*\) has a lifetime of only \(3\times 10^{-16}\,\)s before it fissions. In order for fusion to proceed, a rapid collision with a third nucleus is needed to form an unstable nucleus of carbon-12,
\(\rm {^8Be}^* + {^4He} \buildrel\leftarrow\over\rightarrow {^{12}C}^*,\)
again an endothermic reaction. Energy is finally recovered when the \(^{12}\rm C^*\) nucleus emits two photons to reach the ground state of \(^{12}\)C,
\(\rm {^{12}C}^* \rightarrow {^{12}C} + 2\gamma.\)
Once enough carbon has been created by the triple-\(\alpha\) reaction further alpha captures by \(^{12}\)C can begin. The reaction
\(\rm {^{12}C} + {^4He} \rightarrow {^{16}O} + \gamma,\)
being only a two-particle reaction, proceeds relatively rapidly and
\(\rm {^{16}O} + {^4He} \rightarrow {^{20}Ne} + \gamma,\)
runs more slowly. By the end of central helium burning the composition of a stellar core is typically only 20% carbon with about 80% oxygen along with heavier isotopes.
Nitrogen that has accumulated during CNO hydrogen burning can also capture an \(\alpha\)-particle.
\(\rm {^{14}N} + {^4He} \rightarrow {^{18}F} + \gamma\)
\(\rm {^{18}F} \rightarrow {^{18}O} + e^+ + \nu_{\rm e}\)
\(\rm {^{18}O} + {^4He} \rightarrow {^{22}Ne} + \gamma.\)
Advanced burning stages#
Not much energy per nucleon is available from fusion reactions beyond \(^{16}\)O. Each significant burning stage liberates less energy per nucleon and requires a higher temperature so they last for progressively shorter fractions of a star’s lifetime. Once helium fuel is exhausted the next available fuel is carbon, at about \(5\times 10^8\,\)K,
\(\rm {^{12}C} + {^{12}C} \rightarrow {^{20}Ne} + {^4He}.\)
As the temperature rises photons in the high-energy tail of the radiation field can begin to penetrate nuclei and a new type of reaction, photodisintegration becomes possible. The first major nucleus to succumb is \(^{20}\)Ne. At about \(10^9\,\)K a photon can eject an \(\alpha\)-particle from a \(^{20}\)Ne nucleus. That \(\alpha\)-particle can then be captured by another neon nucleus to form \(^{24}\)Mg. Though the first step is endothermic the combined result is overall exothermic
\(\rm {^{20}Ne} + \gamma \rightarrow {^{16}O} + {^{4}He}\)
\(\rm {^{20}Ne} + {^4He} \rightarrow {^{24}Mg} + \gamma.\)
A little hotter (\(T\approx 2\times 10^9\,\)K) and oxygen can fuse with itself creating mostly silicon-28
\(\rm {^{16}O} + {^{16}O} \rightarrow {^{28}Si} + {^{4}He}\)
Further burning proceeds by a series of photodisintegrations and \(\alpha\) captures. The photodisintegration of \(^{28}\)Si is a limiting step and once it begins, at about \(3\times 10^9\,\)K, burning proceeds to predominantly \(^{56}\)Ni within a day or so. The actual isotopic composition of the iron-group elements, Ni, Co and Fe, as well as V, Cr and Mn, is set by nuclear statistical equilibrium and depends on the ratio of neutrons to protons present.
\(\rm {^{28}Si} + \gamma \rightarrow {^{24}Mg} + {^4He}\)
\(\rm {^{28}Si} + {^4He} \rightarrow {^{32}S} + \gamma\)
\(\rm \dots \rightarrow {^{56}Ni}.\)
The formation of a core of iron-group elements marks the end of a star’s nuclear burning life. No more energy is available from nuclear fusion to support the core which generally has a mass in excess of the Chandrasekhar limit and so collapses to a neutron star or black hole. The enormous release of gravitational energy causes an explosion visible as a supernova which returns the processed outer layers to the interstellar medium.
In the picture so far, helium burning and photodisintegration produce the \(\alpha\)-elements with atomic numbers increasing by two and atomic weights by four. Typically the have equal numbers of protons and neutrons though such configurations can be unstable in heavier nuclei. For instance titanium-44 captures and electron to form scandium-44 that loses a positron to end up as calcium-44. This neutron-rich isotope is second most abundant after calcium-40. The most abundant isotope of titanium is the neutron-rich \({^{48}Ti}\) itself a decay product of chromium-48. Intermediate elements, such as sodium, phosphorus and potassium are rarer and typically require hot hydrogen burning cycles or neutron capture processes.
Type Ia Supernovae and Iron#
Much of the iron formed in massive stars remains in their neutron star remnants rather than being returned to the interstellar medium. So iron is predominantly created by a much rarer phenomenon that involves close binary star evolution.
Luminous type~Ia supernovae are amongst the brightest objects in the Universe. The energy source of type~Ia supernovae is the decay of \(^{56}\)Ni to \(^{56}\)Fe and the total energy released in a type~Ia supernova is consistent with the decay of approximately a solar mass of \(^{56}\)Ni. These facts strongly implicate the thermonuclear explosion of a CO white dwarf though the actual explosion mechanism is not fully understood. In binary systems, mass transfer can increase the mass of a white dwarf. Close to the Chandrasekhar mass (\(M_{\rm Ch}\approx 1.44\,M_\odot\)) degeneracy pressure can no longer support the star which collapses releasing its gravitational energy. Just before this, at about \(1.38\,M_\odot\), cool carbon–oxygen white dwarfs reach densities at which cold carbon fusion begins degenerately and sets off a thermonuclear runaway in which most of the star reaches nuclear statistical equilibrium before entirely exploding.
Elements Heavier than Iron, Neutron Capture#
Neutrons have no electric charge and so no Coulomb barrier to overcome when penetrating a nucleus. They have very high reaction cross-sections with nuclei that increase with the typical area of the nucleus. So neutrons are more easily captured by iron and heavier elements than small light nuclei such as protons and \(\alpha\)-particles. In this way heavy, neutron-rich isotopes can be built up. Consider a general nucleus \(^A_Z\)S, with atomic number \(Z\) and atomic weight \(A\) that captures a neutron,
\(^A_Z{\rm S} + n \rightarrow {^{A+1}_Z{\rm S}} + \gamma.\)
With a slightly larger cross-section the new nucleus can capture another neutron,
\(^{A+1}_Z{\rm S} + n\rightarrow {^{A+2}_Z{\rm S}} + \gamma\)
and similar neutron captures can continue until the new nucleus is so neutron-rich that it is unstable to a weak \(\beta\)-decay in which a neutron emits an electron and a neutrino to become a proton,
\(^{A+2}_Z{\rm S} \rightarrow {^{A+2}_{Z+1}{\rm S'}} + e^{-} + \bar{\nu_e}.\)
Now \({\rm S}'\) is a different element with one more proton than S. Whether or not the \(\beta\)-decay occurs before \(S\) can capture another neutron depends on both the lifetime of the unstable nuclei and the neutron flux. We distinguish the slow neutron capture or \(s\)-process, in which \(\beta\)-decays occur preferentially to further neutron capture, from the rapid neutron capture or \(r\)-process in which neutron captures occur preferentially. Each process generates a distinct set of isotopes in characteristic ratios.
The \(s\)-process#
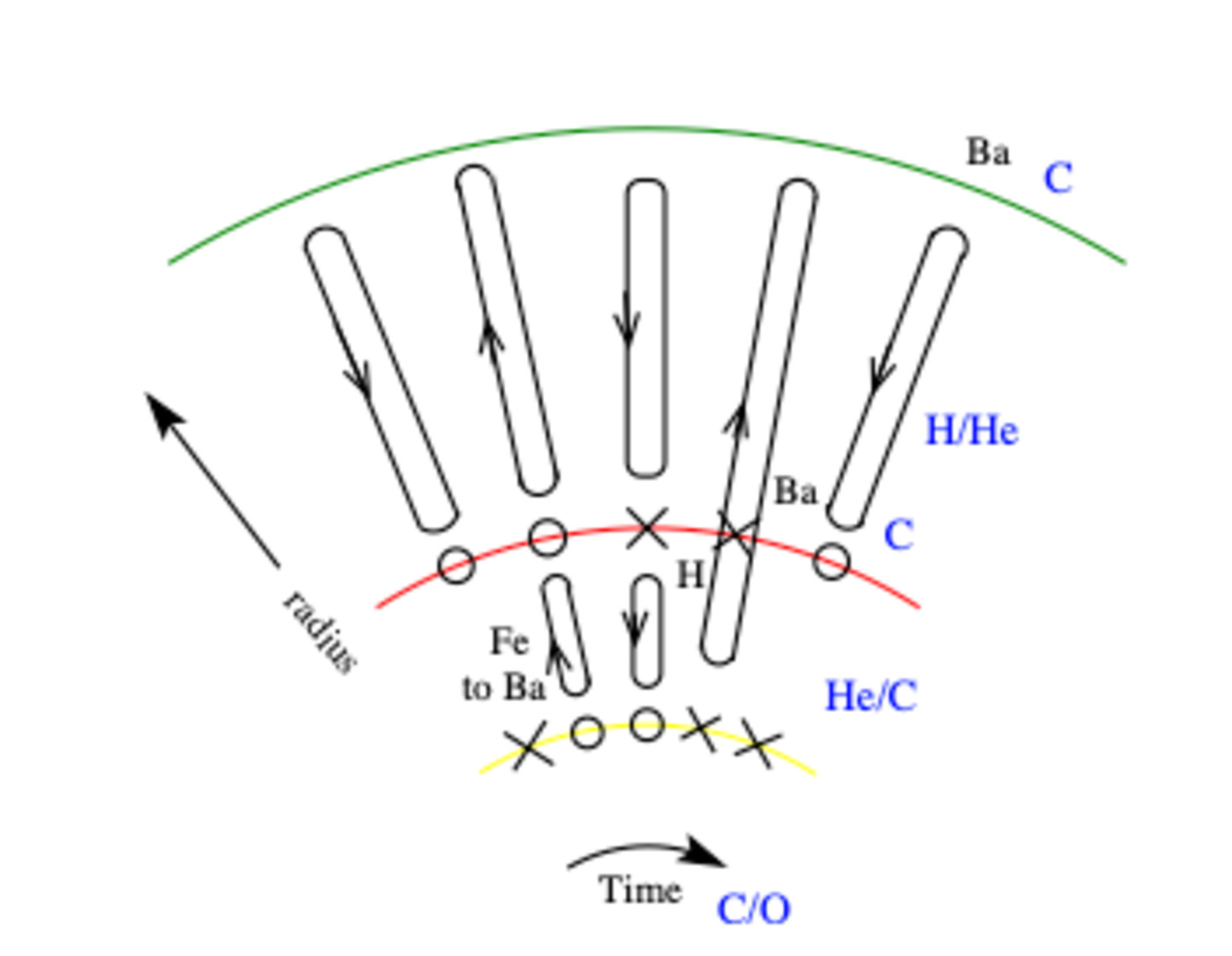
Fig. 2.37 Thermal Pulses on the AGB.#
After second dredge-up in an AGB stars hydrogen and helium burning shells are separated by very little mass. The thin helium burning shell suffers a thermal instability described by H”arm and Schwarzschild in the 1960s.
Interpulse – The helium burning shell is extinguished, the hydrogen shell burns outwards depositing a helium layer on to the carbon-oxygen core for \(10^4\,\)yr after which time the helium burning shell ignites.
On phase – The H”arm–Schwarzschild instability initiates a thermonuclear runaway, for \(10\,\)yr or so, that drives an intershell convective region which quickly removes \(^{12}\)C from the burning shell. The star expands from the inside out so that the hydrogen burning shell cools and is extinguished.
Power down phase – As its fuel runs out the helium burning shell cools over about \(100\,\)yr. The intershell convection ceases and the surface convective envelope deepens through the extinct hydrogen-burning shell into the intershell region. Thence it dredges the products of helium burning to the stellar surface whence it can be returned to the inter stellar medium. Eventually the base of the convective envelope carries fresh hydrogen fuel close to the now extinct helium-burning shell, where the temperature is high enough to reignite shell hydrogen burning and start the next interpulse period.
The \(s\)-process takes place in asymptotic giant branch (AGB) stars during such thermal pulses. The neutron source is either
\(^{13}{\rm C} + {^4{\rm He}} \rightarrow {^{16}{\rm O}} + n\)
or \(^{22}{\rm Ne} + {^4{\rm He}} \rightarrow {^{25}{\rm Mg}} + n.\)
\(^{22}\)Ne is formed during helium burning when \(\alpha\)-particles are captured by the \(^{14}\)N left over from CNO-burning of hydrogen. As a neutron source it requires high temperature helium-burning and so is responsible for neutrons only in massive stars. The CNO-cycle also includes \(^{13}\)C but its equilibrium abundance is much smaller. It can only be produced in sufficient quantities when the number of available protons is similar to the number of available \(^{12}\)C nuclei. This seems to occur in the intershell region of low-mass AGB stars but the mechanism by which the appropriately small number of protons mixes into the intershell region is far from fully understood.
The \(r\)-process#
When the neutron flux is much larger neutrons are captured until nuclei reach the neutron drip line beyond which no more can be accommodated. Once the neutron flux falls off these heavy nuclei rapidly \(\beta\)-decay back to the valley of stability. This \(r\)-process is responsible for elements heavier than \(^{208}\)Pb as well as a characteristic set of lighter isotopes. Europium, molybdenum and uranium are all typical products. The site of the \(r\)-process appears to be merging neutron stars.
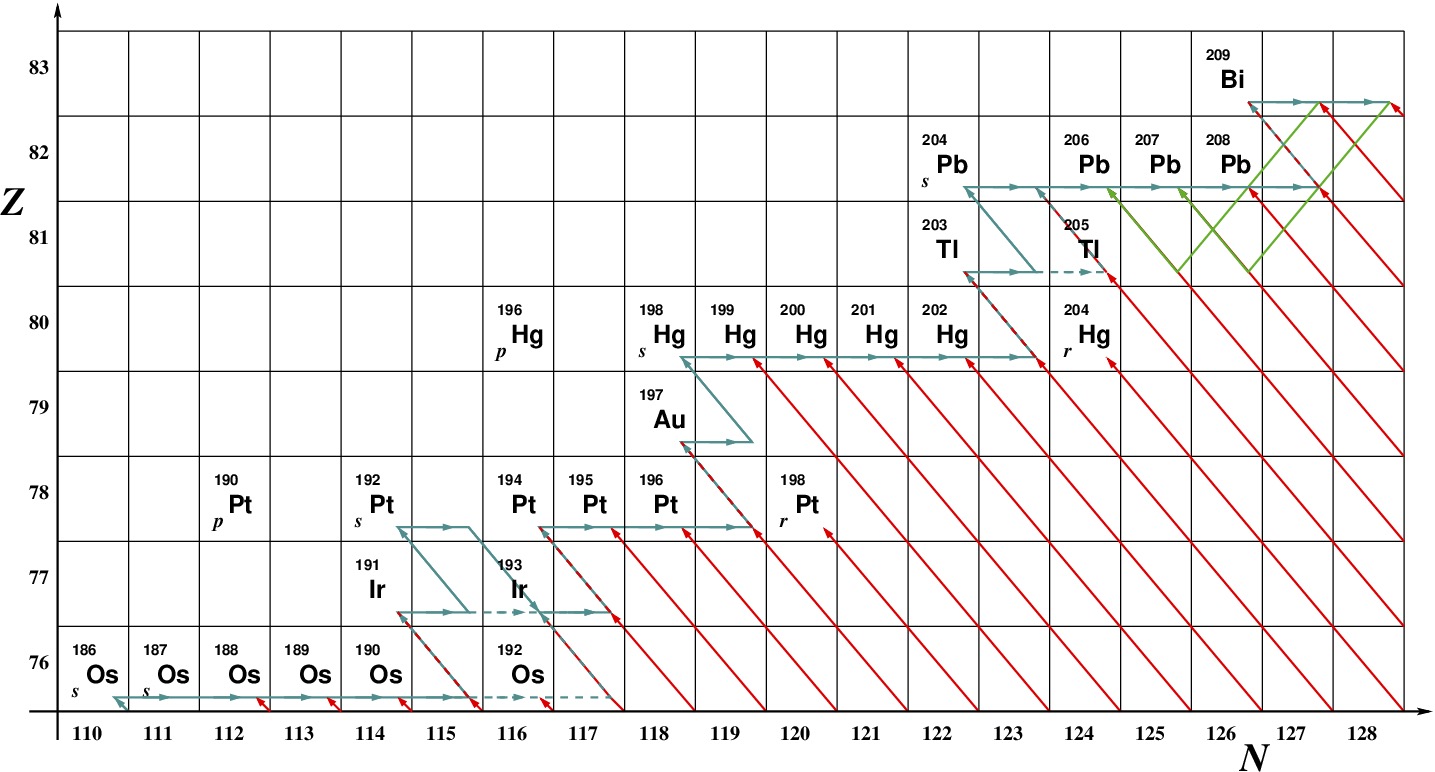
Fig. 2.38 A chart of nuclides in the neutron number–proton number plane for nuclides at the heavy end of the \(s\)-process production. All stable isotopes and those with half lives longer than the age of the Sun are shown. The \(s\)-process path is blue and the \(r\)-process red.#
There are also some proton-rich isotopes that are protected by stable nuclei from both the \(s\)- and \(r\)-processes. These tend to be rare, making up only a tiny fraction of the terrestrial isotopic mix of a given element, and their site of production remains an interesting topic of research. One possibility is Thorne–.Zytkow objects, hypothetical giants with neutron star cores. Accretion on to the core drives a deep convective envelope the base of which is hot enough for heavy nuclei to capture protons. The resulting nuclei are rapidly whisked back to the surface so the burnining is interupted.
